MDS: Moving Beyond the Transfusion Chair
A Conversation with an MDS Expert on Achieving Transfusion Independence
A lower-risk MDS diagnosis can feel anything but low-impact. The fatigue is real. The appointments pile up. And it can be hard to know whether you’re doing everything you could be doing.
In this program, our very own Stephanie Chuang and MDS expert Dr. Mikkael Sekeres from the University of Miami Sylvester Cancer Center talk through what’s actually changed in treatment, what your test results are telling your doctor, and how to walk into your next appointment with better questions.
Program Topics:
- Know your MDS fingerprint
- Finding the right dose: what the latest research shows
- Setting transfusion independence as the target
- Current treatment options & what’s on the horizon
Program Panel

Partners

Thank you to Blood Cancer United for their partnership. They are the world’s leading nonprofit dedicated to curing blood cancer and improving the lives of patients and their families. With over $2 billion invested in research since 1949, they also provide free support services, including their Clinical Trials Support Center and one-on-one guidance from trained oncology information specialists at no cost.

Thank you to The MDS Foundation, a global nonprofit organization dedicated to improving the lives of those impacted by myelodysplastic syndromes — a rare and often undetected blood cancer — and its related diseases. Through patient support, professional education, research, and advocacy, they empower patients, caregivers, and healthcare professionals with the knowledge and resources needed to navigate MDS with confidence.
Program Sponsor

Thank you to our sponsor for its support of our independent patient education program. The Patient Story retains full editorial control over all content.
This interview has been edited for clarity and length. This is not medical advice. Please consult with your healthcare provider to make informed treatment decisions. The views and opinions expressed in this interview do not necessarily reflect those of The Patient Story.
Move from overwhelmed to empowered as you listen to a leading MDS expert discuss the latest in lower-risk MDS treatments, what’s coming, and what it all means for your care.
Edited by: Katrina Villareal
Introduction
Stephanie Chuang: Hi, everyone. Welcome to this conversation titled, “MDS: Moving Beyond the Transfusion Chair.” My name is Stephanie Chuang and I’m a lymphoma survivor. When I was diagnosed, I remember hitting a wall of questions, so I started The Patient Story to help people like you and me find community and get humanized information at and after diagnosis. We do this through featuring incredible patient voices and video stories, and through educational discussions, which I’m excited about with our incredible guest.
We want to say thank you to our sponsor, Geron, for its support of our educational program and our mission to spread the message of patient and care partner self-advocacy. An important note here: The Patient Story maintains full editorial control of the content. Also, importantly, this is not medical advice. We do hope that you walk away with a better understanding of your diagnosis and treatment options, and that you bring this to your own medical team.
Finally, your voice matters to us. We’re going to be having a conversation, but your opinion is key to how we move forward at The Patient Story and what we create, so we want to know your opinion. How did today’s program go? What did you like? What could we do better? And what other topics would you like us to cover?

I introduce our incredible guest who’s going to lead us through this conversation. Dr. Mikkael Sekeres is the chief of the Division of Hematology and professor of medicine at the University of Miami Sylvester Comprehensive Cancer Center. He’s one of the premier myelodysplastic syndromes (MDS) experts in the world and part of dozens of clinical trials and studies when it comes to this. He’s also a prolific author of more than 400 manuscripts, 600 meeting abstracts, and eight books.
Dr. Mikkael Sekeres: Thank you for having me on your program. It is such important work that you do. Communicating topics like myelodysplastic syndromes and rare cancers, introducing people to them, and demystifying them is important, so thank you for this and for having me.
Myelodysplastic syndromes (MDS): Demystifying an unfortunate name
Stephanie: Dr. Sekeres, you spend all your time in this. How do you introduce yourself to patients when they’re first meeting you, outside of the context of the white coat?
Dr. Sekeres: When I first meet people who have the diagnosis of myelodysplastic syndromes, the very first thing I do is exactly what you’re doing with this program: I try to demystify it. It is an unfortunate name, it doesn’t make any sense, and I try to break it down for people.
I start by saying, “What is it that you understand about your diagnosis?” We did a study a few years ago through the Aplastic Anemia and MDS International Foundation (AAMDSIF), where we asked, “How was MDS first described to you?” The vast majority responded that they were told they had a blood disorder and, boy, was that illuminating.
I chair the medical advisory board for the Aplastic Anemia and MDS International Foundation (AAMDSIF). We realized that we had to revamp the educational materials that we were giving to newly diagnosed people so that we could use some of the scary words that we had avoided using in the past, like cancer and leukemia. So when we had informed discussions and said to them, “We would recommend that you receive chemotherapy or a bone marrow transplant,” they didn’t look at us like we had four heads. “What do you mean by a bone marrow transplant for a blood disorder?”
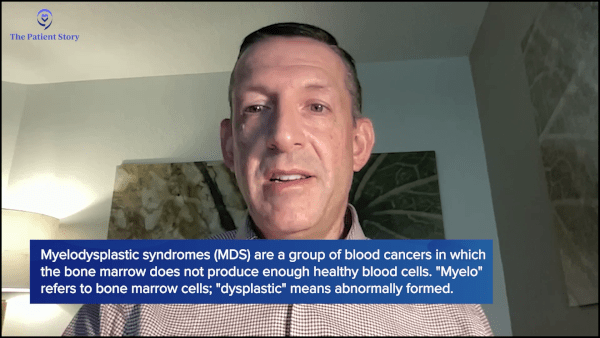
The very first thing we do when we meet is I ask people what they understand about their diagnosis. I break down the term myelodysplastic syndromes to describe the fact that “myelo” refers to the myeloid cells in the bone marrow, and “dysplastic” means bad growing. Literally, it’s describing what we see under a microscope, that somebody has bad growing bone marrow cells, and we take it from there.
Stephanie: That’s wonderful. I love hearing that. You make sure to understand from their perspective where they are and then you address them. “You’ve been told you have this diagnosis. Let’s break down what this actually means.”

Lower-risk MDS vs. higher-risk MDS and what it means for you
Stephanie: What is the next area you go into to help them understand? And then how do you describe how your treatment path is going to run?
Dr. Sekeres: The first thing we do is talk about risk in myelodysplastic syndromes. I use the word cancer with my patients, but I try to put it in context. People who have lower-risk myelodysplastic syndromes (LR-MDS) are going to be living with this diagnosis for years. Some of my patients don’t even require any therapy.
There are people I’ve followed for over a decade. I have never given them a drop of therapy, but we check in every so often. They complain about the traffic when driving in from Miami. I catch up with their family. We look at their blood counts and when we don’t see any change, we schedule the next visit in three, four, or six months, and sometimes even a year. For those folks, I try to say, “We’re giving you a cancer diagnosis,” but I call out the elephant in the room, “But now I’m talking about not even treating you for cancer. How can we even propose such a crazy thing?” And we talk about cancer as a chronic disease.

Other patients have a much more advanced form of myelodysplastic syndromes, something we refer to as higher-risk MDS (HR-MDS), where it does approach the seriousness of having a diagnosis of leukemia. For those folks, we say, “Listen. This is something we need to stop in its tracks. We need to take care of it now, so we’re going to start to talk about therapy. We may even talk about something as dramatic as a bone marrow transplant because that’s the only cure I can offer you for your diagnosis.” A lot of it is contextualizing it and talking about the seriousness of the diagnosis, what we need to do about it, and how quickly.
Can my lower-risk MDS develop into higher-risk MDS?
Stephanie: As a patient, one of the first thoughts is, “If I’ve been diagnosed lower-risk, what is the risk that I’m going to develop into a higher-risk category?”
Dr. Sekeres: That question is a prescient one and probably the most important thing that people think about immediately. It’s something where we don’t have a tremendous amount of data to guide people. People can evolve into a higher-risk MDS (HR-MDS) or even acute leukemia from lower-risk MDS (LR-MDS), and that can take months, years, or even over a decade.
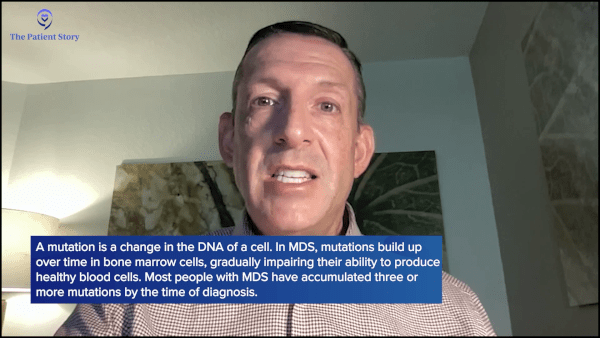
We learned that a cell gets a mutation and then you get cancer, but it’s a lot more complicated than that. When a cell gets a mutation, you might live with that mutation for years, not even knowing that you have a problem, and then that cell gets another mutation and another mutation. Typically, at diagnosis, people with myelodysplastic syndrome have three mutations, so it takes three or more mutations for that cell to change enough that the blood counts start to become compromised, which is when we first diagnose myelodysplastic syndrome.
When I meet them, they may only have one mutation. Some of my patients may only have two mutations. We can’t predict how long it’s going to take for them to acquire a third or fourth mutation that would start getting them into trouble. But they realize that MDS is something that can occur and develop over years, decades, or even an entire life.
Stephanie: What is the ballpark breakdown of how many patients who come into a doctor’s office with an MDS diagnosis fall into the lower-risk category versus the higher-risk category?

Dr. Sekeres: This is very approximate. Roughly three-quarters of patients have lower-risk MDS (LR-MDS). About one-quarter to one-third have higher-risk myelodysplastic syndromes (HR-MDS). For those two-thirds or three-quarters who have lower-risk MDS (LR-MDS), we may not be talking about any therapy at all. We may just monitor them a little more frequently in the first year — maybe once a month, every two months, every three months, or every four months.
And then, if we can demonstrate that things are stable with our blood counts, we spread out how frequently we monitor them. We go every three months, four months, or six months. I even have patients whom I monitor once a year. My former patients in Cleveland are snowbirds. They come down to Florida and make a part of the trip to see their MDS doctor for their blood counts.
Stephanie: Great. They moved with you. They’re like, “Great! Now we have a reason to go to the sunshine when it’s cold over here.”
Dr. Sekeres: It’s so interesting. People make more of an excuse to come to Miami to visit me than they did when I was in Cleveland.
Stephanie: Amazing. That’s a great plug for Sylvester Comprehensive Cancer Center in terms of the benefits of going there.
What signals should patients watch out for?
Stephanie: Dr. Sekeres, this program is focused on lower-risk MDS (LR-MDS) because the treatment experience is very different in terms of how it’s managed. It’s slow-growing, slow-moving. How do you introduce — ballpark — what people should be looking out for, how they should be talking to you, and how often they should see you?
Dr. Sekeres: A way to introduce this is to think about goals of care for lower-risk myelodysplastic syndromes (LR-MDS). With some patients, I just follow their blood counts over time and we don’t intervene at all. No study in MDS has ever shown that intervening early with a therapy — before somebody has symptoms or a compromise in their blood counts or quality of life — allows somebody to live longer than when we wait before we start therapy. That is a truism in certain types of cancers where we don’t treat people immediately.
There was a very famous study of people who have chronic lymphocytic leukemia (CLL), which is analogous to MDS, where sometimes people are diagnosed at a very early stage. They have a slightly elevated white count, but otherwise, they’re great. It was picked up by accident. There was a study that randomized those people to receive treatment immediately or to wait until their blood counts started to suffer or they started to have symptoms.
The goals of therapy for lower-risk MDS (LR-MDS) are to improve quality of life or improve blood counts, which is another way of saying minimizing or eliminating any requirements for blood or platelet transfusions. If my patients have a good quality of life and don’t need any transfusion, there’s no reason to start therapy.
But once that changes, once my patients start to tell me, “I used to be able to walk two miles a day, but now I’m down to a couple of blocks because I get so short of breath or I get so tired,” or they start to need a blood transfusion every eight weeks, and then it becomes every six weeks, and then every four weeks, that’s when we start to talk about intervening with treatment to improve their quality of life.
Stephanie: I love that you’re talking about this because it gives real-world examples of when you might be looking at signals of a shift. Are there any other ones that are more common that they should be aware of? Anything that would signal, “Hey, I should tell Dr. Sekeres about this because it could have real implications as to when to start treatment.”
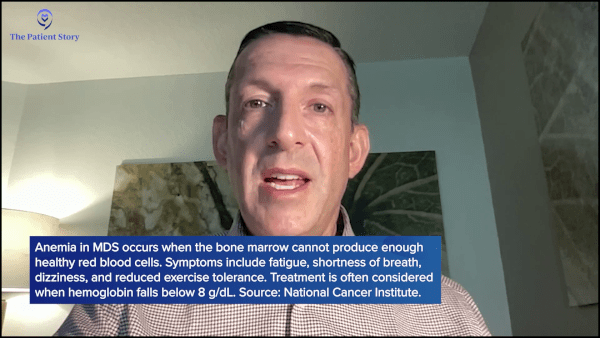
Dr. Sekeres: Over 80% of people with myelodysplastic syndromes have some degree of anemia, so we focus a lot on the signs of anemia. A dropping hemoglobin or hematocrit is a sign of anemia. Once that drops below 10 or nine and heading towards eight or lower than eight, we start to talk about initiating therapy. The vast majority of my patients don’t have major symptoms until their hemoglobin gets less than eight. There are exceptions to that, though. Sometimes people with severe heart disease will start to get symptoms with a hemoglobin level of less than 10.
The only way we can tell if fatigue is due to MDS or not is by giving a blood transfusion. If, after a blood transfusion, the patient says, “I feel like Superman or Wonder Woman for the first time in months,” then we know that the low hemoglobin was causing their fatigue.
But, Stephanie, most of my patients are older. When I’m talking to somebody in his or her 70s or 80s and they say to me, “Oh, I feel tired,” it’s hard to discern whether that’s, “I feel tired from my MDS,” or “I feel tired because I expect myself to feel like I did when I was in my 30s and I’m not in my 30s anymore.” We try to get to the truth with fatigue in people with myelodysplastic syndromes: whether it’s due to the diagnosis, other conditions like heart disease or lung disease, or comparing how you feel at age 85 to how you felt at age 35, and whether starting a treatment will help.

Dr. Sekeres: Fatigue can be due to anemia. Shortness of breath can be due to anemia. Some of my patients will say their appetite is down because of anemia. More infrequently, people will start to get chest pains due to anemia if they have underlying heart disease, because anemia is a form of a stress test for the heart. I pay attention to those symptoms as well.
Outside of symptoms related to anemia, if somebody has a low platelet count, they may be more prone to bruising and to developing a rash called petechiae. It looks like someone took a stick pin to their skin, but it doesn’t bleed; it’s bleeding underneath the skin. Or frank bleeding; we’re talking about nosebleeds, bleeding when you brush your teeth, or blood in the urine or stool. Those are definitely signs that we need to check the platelet count and may need to start therapy.

The final blood count we focus on is the white blood cell count, which can also go down when someone has myelodysplastic syndromes. The WBC is a marker of our immune function. Patients may start to get recurrent infections, which can be random infections. I had one patient who always got an infection of his earlobe and I have no idea why. Another person gets recurrent urinary tract infections (UTIs). That’s another signal that we may need to start therapy to try to correct the blood counts and minimize the chance of infections.
Stephanie: I know it’s a lot to consider, but as you said, a lot of MDS patients, especially those who are older, may not understand when to mention something that they’re feeling because they may say that’s how it normally is. What’s your message for people when in doubt if they’re feeling anything at all? How often should they be reporting anything that they’re feeling?
Dr. Sekeres: It’s always important to bring up with your doctor if you have a symptom. It may or may not be related to an underlying bone marrow cancer, like myelodysplastic syndromes. We can help figure that out. It may take a what I refer to as a litmus test of giving a blood transfusion, for example, and seeing if that symptom goes away or not, and being judicious about starting therapy. Not every symptom is related to myelodysplastic syndromes, but let us help you figure it out.
Stephanie: I like that message. It’s a team effort, obviously, especially with this long-term relationship.

Understanding your MDS risk score and using the IPSS-M calculator
Stephanie: I would be remiss not to ask about mapping the MDS fingerprint, so to speak, and demystifying some of the alphabet soup a little bit. There is something called the IPSS-M related to mutations, which might come up in the conversation. How does that come up and what should patients understand?
Dr. Sekeres: One of the questions I often get is, “I have a sister who had breast cancer and she was stage 2,” or “I have a cousin who had lung cancer and he was stage 3. If this is cancer, what’s my cancer stage?” The challenge with staging systems is that they’re designed for people who have solid tumors, like breast cancer, lung cancer, prostate cancer, and colon cancer. They don’t work very well for people who have cancers of the bone marrow.

Back in 1997, the very first International Prognostic Scoring System (IPSS) was published. What it did was to codify what is pretty obvious if you have myelodysplastic syndrome or you’re somebody who treats myelodysplastic syndrome. People who have multiple low blood counts or multiple cytopenia, those who have particularly poor risk genetic mutations in their MDS cells, and those patients who have very high blast percentages.
Blasts are immature white blood cells in the bone marrow. We all have blasts. Stephanie, you and I have 1% or 2% blasts. That doesn’t mean that we have anything serious. It means that we have normal, immature white blood cells that are making our mature immune system. But once you get 5% blasts, we say that’s abnormal. Once you get 20% blasts, we switch the diagnosis from myelodysplastic syndrome to acute myeloid leukemia.
The IPSS-M, which is the International Prognostic Scoring System-Molecular, gets to the nitty-gritty of those genetic mutations. It’s much more sophisticated at adding them up. It’s become so complicated that I can’t do it on the back of an envelope anymore. I have to enter my patient’s clinical data onto an online website. But you know what? As someone with MDS or a family member of MDS, you can enter your own data or your family member’s data, your friend’s data directly into that website, and it will kick out a score.
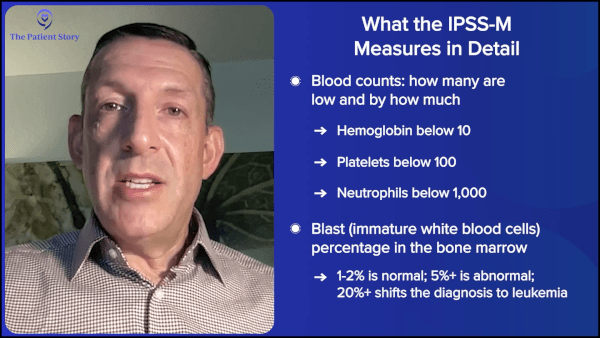
The second thing we focus on when we come up with a prognosis is the blast percentage you have in your bone marrow. Either 1 or 2% is considered normal. Once you get to 3 or 4%, it’s a little bit abnormal. Once you get to 5% or higher, it’s more abnormal. And once you reach 20%, we then shift the diagnosis to acute myeloid leukemia.

Finally, we talk about the genetic mutations in your MDS cells. There are two ways that we measure that. One is something called cytogenetics, where we literally look at the 23 pairs of chromosomes in the myelodysplastic syndrome cells and look for abnormalities, so that’s a global view. Then we look at the specific mutations that we’ve already discovered are linked to myelodysplastic syndrome and we check if one of those has one of these mutations. That’s referred to as next-generation sequencing (NGS); another way of referring to that is molecular mutations. You get all of that information and that’s the information that we feed into the IPSS-M to generate a score and a prognosis.
There are a couple of caveats to all of that. The prognosis that the IPSS-M kicks out is for a huge population of people. It doesn’t necessarily reflect your prognosis. I frequently will walk my patients through all of this, Stephanie, and at the end, they’ll say, “Oh, so you’re saying I’m going to live for 23 months?” No, that’s not at all what I’m saying. You may live for years, but a typical person with your characteristics is expected to live for 23 months. It’s an average. There are just as many people who live less than 23 months as there are who live more than 23 months.
What the prognostic system doesn’t feed into the equation are some of the intangibles. How is your health otherwise? If you don’t have heart disease, lung disease, or kidney disease and the only wrong thing is the MDS, your prognosis is going to be better.
These prognostic systems also don’t incorporate your social supports. If I walk into a room and I have a bunch of family members who are in there with the person who has the diagnosis of MDS right off the bat, I know that their prognosis is going to be better because they have the support at home, as opposed to somebody who comes all alone to my clinic, lives alone, and doesn’t have the support system. That’s someone who’s going to have a rougher time with it.
Stephanie: Again, I appreciate the nuance you’re bringing into this because, of course, one of the first questions for people is prognosis and how long they have. When you’re diagnosed, you see the averages and don’t know where they come from, and you’re very literal about that. I appreciate the encouragement to talk more about this with your medical team and understand what it means on a personalized basis.
What your gene mutations mean for your risk and treatment decisions
Stephanie: How does mutation show up in the treatment decision-making process?
Dr. Sekeres: We use mutations in a couple of different ways. One is how it feeds into prognosis and whether we believe somebody has a lower-risk MDS (LR-MDS) or a higher-risk MDS (HR-MDS). Some of those mutations are poor-risk mutations. If we look at populations of people who have MDS with those mutations in the past, the therapy doesn’t work as well, and they may not live as long. For those folks, we’re going to encourage them to be more aggressive about treating their MDS. For certain folks, we’re even going to recommend a bone marrow transplant as we recognize that’s the only chance for a cure. We use molecular mutations to help modify risk, how we talk about risk, and how we talk about the risk of treatments.

Rarely, somebody will have a myelodysplastic syndrome with a mutation that we can actually target with a drug. This doesn’t happen as often as we would like, but some people have IDH mutations. We have three drugs on the market that target IDH mutations, which may be part of the treatment immediately or eventually. Very rarely, we’ll have someone who has an FLT3 mutation, and there are three drugs on the market as well that target FLT3 mutations. More commonly, we will see people who have an SF3B1 mutation, also known as a spliceosome mutation.

You’ll notice that all of these mutations are gobbledygook of letters and numbers, so you can’t make a lot of sense of them when you hear them. You have to write them down and then discuss with your doctor, nurse, or pharmacist. You can look it up online to know how it affects treatment recommendations, but allow your healthcare provider to contextualize what you find online because it can get awfully confusing very quickly.
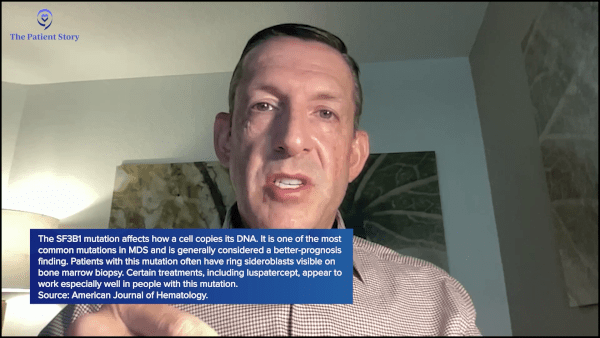
Stephanie: Can we use the more common one, SF3B1, as an example for how you might communicate with a patient about why this would matter? How do you describe the significance of knowing that with a patient who has that?
Dr. Sekeres: An SF3B1 mutation, also known as a spliceosome mutation, is a mutation in how when a cell divides and makes a copy of itself, it also has to make a copy of its own genetics, its own DNA, and to do that, a cell has to unravel the DNA. Remember, the DNA is one of those spiral staircases, right? It wraps around itself. The cell has to untwist, cut it open, and allow RNA to get in there and copy the genetic sequence to make a whole new strand of DNA, a whole new chromosome.
In doing that, it has to make snips or splices, so that you can actually have a mutation in the thing that makes the cuts of the DNA: spliceosome. That’s what an SF3B1 mutation is. It’s associated with people who have MDS with what are called ring sideroblasts (RS).
If you look at your bone marrow report and see a pathologist comment on the presence of ring sideroblasts (RS), what that means is when we take a bone marrow sample and put it on a slide, pathologists will then add a dye to that slide, and some of the cells will take up that dye in a ring, like Saturn has rings around it. Some of your cells will have a ring around them. Those rings are much more common in people who have an SF3B1 mutation.
It’s actually a good prognosis mutation to have. People who have an isolated SF3B1 mutation live longer than people who have other types of mutations. More recently, we’ve recognized that there are some therapies that seem to work better in people who have the SF3B1 mutation than in people who don’t have that mutation.
First-line treatment options for lower-risk MDS (LR-MDS)
Stephanie: As you talk with patients about their options, you talk about signals for when you might start treatment. How do you assess the first-line treatment options for patients and communicate that to them in terms of the why in a digestible way?
Dr. Sekeres: We talked a little bit earlier about what hemoglobin level triggers us to start to recommend therapy. For most of my patients, that’s a hemoglobin level of eight. Once their hemoglobin starts to head towards eight or dips below eight, that’s when we know that we’re going to need to start therapy.
Now that could be a blood transfusion. I have some patients who get one blood transfusion every three or four months. That doesn’t have a major impact on their quality of life if they’re only getting blood four times a year; that isn’t too bad. They’re willing to continue to get a blood transfusion with that frequency because it doesn’t interrupt the other things that they want to do.
Once the frequency starts to increase to once per month, it starts to become a hassle. Now they have to deal with the traffic in and around Miami. They have to park. They have to remember to bring some cash to tip the valet guy. They need to check in. They need to have an appointment with the doctor and an appointment for a lab draw. They need to go to an infusion center. They need to sit for the infusion. Then, after, they need to collect their car and drive home.
You add all of that time up and you’re talking about a full day to get a blood transfusion. A full day every four weeks starts to add up, which is when my patients start to say to me, “This MDS is starting to get annoying. Is there any treatment that I can take to start to eliminate the frequency of blood transfusions?”
In general, the trigger is time or the hemoglobin level. Occasionally, my patients will start to complain of progressive fatigue and that’s when we start to try to figure out whether the fatigue is related to the MDS or something else. If it’s related to the MDS, we need to start a treatment to improve their hemoglobin level, so my patients don’t get as fatigued.
Stephanie: What are the most common first-line treatment options that you recommend for patients and how do you make your decision on what to recommend?
Dr. Sekeres: We start with the treatment that has the most likelihood of working and the fewest side effects, so it’s the relative balance of safety and efficacy.
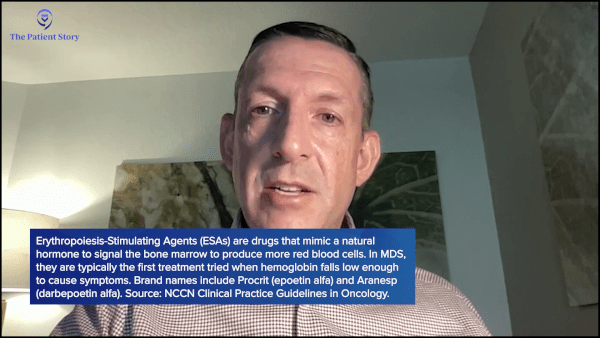
For most people, that represents an erythropoiesis-stimulating agent (ESA). It’s a hormone that stimulates the cells in the bone marrow to make more red blood cells. People who have kidney disease are treated with the same hormone, so it’s not a form of chemotherapy. Doctors around the country are used to giving it, so you don’t require a specialty center to give one of these drugs.
The hormone is referred to as either erythropoietin (EPO) or darbepoetin, and it can be given once a week or as infrequently as once every three weeks. It works about 30 to 40% of the time. If I’m über simplifying it, I would say about one-third of my patients have an improvement in their hemoglobin level or even an elimination of the need for red blood cell transfusions with this drug. On average, it’s effective for somewhere between a year and two years.

Stephanie: Are there other treatment options that you present? You said that was one-third of the folks. For the rest of the patients who don’t see that, what is the conversation with them?
Dr. Sekeres: When someone’s hemoglobin level is low, when they start to need transfusions or are about to need transfusions, we will usually have a conversation about whether they should go on an erythropoiesis-stimulating agent (ESA), like erythropoietin or darbepoetin, or whether they should consider treatment with the drug luspatercept (Reblozyl). Whereas erythropoietin and darbepoetin work on early stages of growing red blood cells, luspatercept (Reblozyl) works on later stages of growing red blood cells, and it seems to have particular efficacy in people who have the SF3B1 mutation or ring sideroblasts (RS) in their bone marrow.

There was a study that randomized people with lower-risk myelodysplastic syndrome (LR-MDS) who were already dependent on red blood cell transfusions to receive either erythropoietin or luspatercept (Reblozyl). In the entire study, about 60% of people who received luspatercept (Reblozyl) went at least 12 weeks without needing a blood transfusion anymore, so that was what was considered a success and effective. That was compared to about 31% of people who were initially treated with erythropoietin.
However, when you break down that population of people to those who have the SF3B1 mutation or ring sideroblasts (RS) versus those who don’t, with those who don’t have the SF3B1 mutation or ring sideroblasts (RS), there was no difference in efficacy between those who received luspatercept (Reblozyl) or erythropoietin. They were roughly equivalent.
However, for those who did have the SF3B1 mutation or ring sideroblasts (RS) in their bone marrow, the luspatercept (Reblozyl) worked significantly better than erythropoietin or darbepoetin. The discussion we usually have is if you have the SF3B1 mutation or ring sideroblasts (RS) in the cells in your bone marrow, in general, we will favor starting with luspatercept (Reblozyl) because it’s going to be more effective and for a longer period of time.
If you don’t have the SF3B1 mutation or ring sideroblasts (RS), we will say dealer’s choice: either works about the same. If your doctor is more comfortable giving erythropoietin or darbepoetin, then it’s totally fine to get that upfront.

There is a drug called lenalidomide (Revlimid) that is specific for treating patients who have a deletion 5q mutation. We talked about the specific next-generation sequencing molecular mutations and the big chromosome mutations. The big chromosome mutation is a loss of a portion of chromosome 5 called deletion 5q.

For patients who have that mutation, a form of targeted therapy is to receive lenalidomide (Revlimid). About two-thirds of people with that mutation will get better when they get lenalidomide (Revlimid) for an average of about two years. If you have that specific mutation, that is a great drug upfront as well.
Second-line treatment option for lower-risk MDS (LR-MDS)
Stephanie: This is part of why we talk about information about our own disease and what’s going on, because that does inform the treatment choices that would come up. There’s also one that came up as potentially disease-modifying: imetelstat (Rytelo). Can you talk about that also and when you might talk about that with patients?

Dr. Sekeres: Imetelstat has been approved by the FDA for people who have lower-risk myelodysplastic syndrome (LR-MDS) where they’ve already received blood transfusions and been exposed to an erythropoiesis-stimulating agent (ESA), either erythropoietin or darbepoetin, and the drug either didn’t work or stopped working. In the study that led to its approval, about 40% of people had an improvement in their transfusion needs, so they went from needing blood transfusions regularly to no longer needing blood transfusions. And on average, the drug worked for about a year.
The drug works on telomerase. It’s complicated. It has to do with snipping the ends of DNA and how important that is. The drug works in people who’ve been treated previously with other drugs. We did present some results that showed that it works as well after people have received just one of the erythropoiesis-stimulating agents (ESAs), as in patients who’ve already received lenalidomide (Revlimid), for people who have a deletion 5q mutation. It also works in people who’ve already received luspatercept (Reblozyl), but probably doesn’t work as well in people who have already received azacitidine (Onureg, Vidaza) or decitabine (Dacogen).
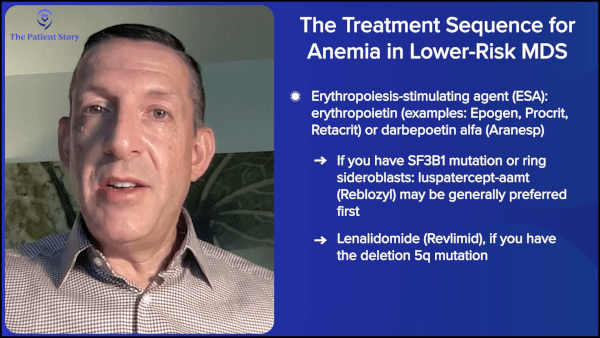
The order in which we think about using drugs for people who have anemia with lower-risk MDS is to start with an erythropoiesis-stimulating agent (ESA). If they don’t have ring sideroblasts (RS), start with luspatercept (Reblozyl). If they do or have the SF3B1 mutation, then move to lenalidomide (Revlimid). For people who have the deletion 5q mutation and then move to imetelstat (Rytelo) before moving to azacitidine (Onureg, Vidaza) or decitabine (Dacogen).
Does any of this modify the disease?
Stephanie: We’ve heard about supportive care. There are symptoms and trying to manage the quality of life. Then there’s the question of whether this acts to help with addressing the disease or the production of the bad cells. Can you talk about that and how that factors in with conversations about therapy and treatment decisions?
Dr. Sekeres: That’s a great question, Stephanie. You would think that we would be focusing on either supportive care or modifying the disease, but in truth, we don’t. We focus more on the blood counts and the order in which we offer medications based on their relative balance of how effective they are, how well they work versus the side effects, and how safe they are. That’s why we’ll go in the order that I mentioned. It’s a mix of things that would be considered supportive care versus disease-modifying.

To be completely blunt, the reason we do that is that we’re not sure how much it truly modifies the disease. None of these drugs in a prospective study — where at the very outset, in a randomized trial, the goal was looking at overall survival — has been shown to improve how long people live. They’ve demonstrated that they modify symptoms and improve quality of life; we published that with the imetelstat (Rytelo) data, but they haven’t shown in a randomized phase 3 trial that they allow one group of patients to live longer than another group of patients.
And that’s why we started by talking about goals of care with lower-risk MDS. The goals of care are to minimize transfusions and improve quality of life, and that’s what we can do with these drugs. Erythropoiesis-stimulating agents (ESAs) may be thought of as supportive care, and the others may be thought of as more than that. Or maybe luspatercept (Reblozyl) is lumped into those that are thought of as supportive care, and the others “modify disease,” but none of them modifies disease enough — that we know of — to allow somebody to live longer.
Stephanie: I know we don’t have a crystal ball, but we have heard questions about disease modification. What does that mean? This is how we’ve been treating the disease. It could be that way for a while, but here’s where we could go as we’re starting to talk about different mechanisms of action.

Dr. Sekeres: Our goal is to modify disease so that people live longer and live better, and that goal is aligned with the FDA with what they’re trying to do when they approve drugs. We recognize that these trials may be designed with short-term endpoints to get a drug approved quicker, but it may very well be that these drugs — particularly the ones that modify disease like imetelstat (Rytelo), azacitidine (Onureg, Vidaza), and decitabine (Dacogen), and the oral decitabine combined with cedazuridine (Inqovi) — may be modifying disease enough that people are actually living longer, but we haven’t captured that yet in a rigorous trial design. So that may be happening. We may be achieving this goal without even realizing that we’re achieving this goal.
MDS is a very complicated, very heterogeneous collection of diagnoses. There are panels of gene mutations that have 25, 50, 60, 75, 100 mutations; that’s how complicated MDS is. And it’s our job to continue to develop drugs that target every one of those mutations to make a dent in a person’s quality of life and their overall survival. That’s the future of this.
Stephanie: I appreciate you describing it and addressing that because it is something that people think about. Where are we headed?
The BRAN framework and advocating for yourself
Stephanie: Before we wrap, I’d like to spend time on the idea of independence and quality of life. You talked about transfusion independence and making sure that there’s lower fatigue and mental clarity. How often do you use the BRAN tool? Is this a good framework for patients to use in terms of thinking about questions they can ask? That’s (B)enefits of new data, (R)isks of specific treatment, (A)lternatives available, and then (N)othing, what happens if we stay the current course.
Dr. Sekeres: That’s a terrific way of thinking about it. I love it when my patients bring in new trial results to me and ask, “What do you think about this?” It actually happened recently. Someone brought in some trial results and, unfortunately, it was a very early phase study, so there’s not much we can make of it. It’s a study that’s enrolled 12 or 20 people so far. My response to those is often, “So far, so good. Let’s see where it goes. Maybe this will be a new type of treatment.”

When you’re assessing the benefits of new data, it’s important to ask: Is this applicable to me right now? Sometimes it is, sometimes it’s not, though. Sometimes it involves a different patient population.
Always ask about the risks of a specific treatment. It’s a balance. Think about it the way I do. What is the relative balance of efficacy of a new drug and the safety of that drug? If the balance of fficacy and safety is in favor of efficacy, then we try it. If it’s not, if it looks like the side effects and the toxicities of that drug are going to outweigh any relative benefits of that drug, then that’s not a route we should go.
Always ask about alternatives. What other therapies could I try in place of this?
And then if we stay on the current course, what is our goal here? That’s a great opportunity to talk with your doctor or nurse about your goals of therapy and make sure that they’re aligned with the goals of your healthcare team.
New treatments and approaches that came out of ASH
Stephanie: We have the American Society of Hematology (ASH) annual meeting, the biggest blood disease, blood disorder, and blood cancer conference every year, where specialists like you get together to talk about the latest. Definitely, our patient and care partner community likes to understand the latest developments coming out of a conference like that and how close we are to seeing the impacts of what’s being discussed. You’ve given a landscape of how you would make treatment decisions. Can you bring us through some of the more impactful updates that came out of ASH most recently and what’s part of the conversation for you as a specialist today?

Dr. Sekeres: Coming out of the most recent American Society of Hematology annual meeting, there’s a new drug called elritercept, which looks like a next-generation luspatercept (Reblozyl). In an initial study in which everyone got the drug, it looked like at least the same number of people, if not more, seemed to get better with elritercept as we would have expected from the same people receiving luspatercept (Reblozyl). That drug is now being explored in a randomized trial, similar to the randomized studies conducted with luspatercept (Reblozyl), to see if it’ll be the next drug approved by the FDA for the treatment of lower-risk myelodysplastic syndromes (LR-MDS).

Another drug that’s being studied uses a new mechanism of action, so it works differently from the other drugs that are out there for myelodysplastic syndromes. It’s what’s referred to as an IRAK inhibitor. In earlier phase studies in people who have already received other treatments for their myelodysplastic syndromes, we’re once again seeing some nice improvements in a percentage of people receiving this drug. That is also in the middle of clinical trials to see whether it will enter a randomized study to get FDA approval eventually.

Finally, we looked at a “less is more” approach to treating people with lower-risk myelodysplastic syndromes (LR-MDS) using drugs that are already on the market. We were focusing on azacitidine (Onureg, Vidaza) and decitabine (Dacogen). These are more commonly used for people who have higher-risk myelodysplastic syndromes (HR-MDS), but they’re also used in people who have lower-risk myelodysplastic syndrome (LR-MDS) who have multiple blood counts that are abnormal — maybe they have anemia and a low platelet count or anemia and a low white blood cell count.
In this study, we gave less of the drugs. Instead of giving five days of decitabine (Dacogen) or seven days of azacitidine (Onureg, Vidaza) — which is the FDA-approved schedule of these drugs and what we use in patients with higher-risk MDS — we used three days of decitabine (Dacogen) or three days or five days of azacitidine (Onureg, Vidaza). We found that less is more. More people had improvements and it looked like people were even living longer with getting less of these drugs for lower-risk MDS.

This aligns with an initiative that the FDA has called Project Optimus. They’re encouraging looking at lower doses and less frequent schedules to see if, in their own approval of these drugs, they may have gone too far, and we can scale it back for people to minimize the side effects that people have to these drugs.

When to see a specialist and why it matters
Stephanie: What’s the importance of seeing a specialist like yourself? When should someone talk about seeing someone who specializes in MDS? It’s not a common diagnosis. And how do you work with the doctors and the hematologist-oncologists who are in the community?
Dr. Sekeres: The way I look at it is that myelodysplastic syndromes are a rare diagnosis. It affects about 20,000 people in the United States each year. Compare that to the number of women diagnosed with breast cancer, which is over 200,000, prostate cancer, which is over 200,000 for men, and lung cancer, which is about 200,000. It’s a rare cancer. You owe it to yourself and your family to go to a specialist, not only for the advice that we may be able to give, but also for the specialist pathologist, the person making the diagnosis.
We did a study through the National Heart, Lung, and Blood Institute (NHLBI) where we looked at diagnoses of MDS that were made at a community site and compared them to the diagnoses that expert pathologists made. We found that there were discrepancies 20% of the time. And this wasn’t some subtle discrepancy where an MDS nerd like me would say, “Does that have enough ring sideroblasts (RS) to count?” These are differences in diagnoses between MDS and leukemia or MDS and no cancer at all. What was scary was that 7% of the time, we found that people received the wrong therapy for the wrong diagnosis. Yikes.

You owe it to yourself not only to go to a specialty center to get a specialist’s opinion on what to do next, but also to get that specialist pathologist to take a look at the bone marrow biopsy results to make sure that you have what you’re told you have.
Self-advocacy and working with your care team
Stephanie: We’re aligned here on the mission of empowering patients with the understanding and the ability to ask questions, so they can make the best decision for themselves. What do you say to those who say this is just what it is and there’s nothing they can do about it? What is your message on the power of being able to self-advocate and know a little bit more about what they’ve been diagnosed with, themselves or their loved ones?

Dr. Sekeres: We all deserve an advocate in our lives, particularly when it comes to health care. Sometimes that advocate can be you yourself. But you know what? Sometimes you don’t feel well because you have a medical diagnosis. Identify an advocate who’s close to you, someone who’s going to speak up on your behalf. That may be a partner you live with, one of your kids, or a good friend. I’ve had all sorts of advocates accompany my patients to their appointments.

Always prepare a list of questions you want to ask before you walk in the door. I’m a doctor, but when I go to my doctor, my mind goes blank. It’s ridiculous. It should never happen, right? But, of course, it does, because you’re in the heat of the moment. You’ve been waiting for a while. You’re debating whether you should go to the bathroom to pee. Are you going to miss the doctor if you leave the room? You’re in this scenario and then, all of a sudden, the doctor and the team walk in. All of a sudden, you’re in the spotlight and you’re asked, “Do you have any questions?” And you forget. Write down your questions beforehand and have an advocate with you who’s going to speak up on your behalf.
Stephanie: Everything you described, I’ve definitely felt before. I also appreciate you humanizing it by saying, “Look, as a doctor, even I sometimes blank out because it’s overwhelming at another doctor’s office,” so thank you for that.
Conclusion
Stephanie: Thank you for joining the program and spending so much time, not just with your own patients and the research, but on programs like this, where we try to distill down what patients and their care partners can take away. Thank you, Dr. Sekeres.
Dr. Sekeres: Stephanie, thanks for being such a great advocate for people.
Dr. Sekeres is from the University of Miami Sylvester Comprehensive Cancer Center. We are thankful for his commitment and for spending the time with us. While he is a top MDS specialist, remember that this program and conversation were for informational purposes only, so please bring your questions and decision-making to your medical team.
Thank you again to our program sponsor, Geron, for supporting our educational program. We want to remind you that The Patient Story maintains full editorial control.
There are a lot of resources. At The Patient Story, we have patient story videos and other educational programs. You can also check us out on YouTube. We have hundreds of videos with patient voices that may be helpful for you, a friend, or a family member. There’s also the MDS Foundation’s IPSS-M Calculator, which helps get your risk score and can help with treatment decision-making. The foundation also has support groups and other patient resources, so visit their website.
I also want to give a shout-out to our friends at Blood Cancer United, which has information specialists you can reach anytime by phone. The Blood Cancer United group also has an online community for peer support.
I’m so glad that you could join us and I hope to see you at another program. For now, from The Patient Story, I’m Stephanie Chuang. Take good care.
Hear from people living with myelodysplastic syndromes
Real experiences with diagnosis, treatment choices, side effects, and life beyond MDS — in their own words.


