-

·
How Does Sugar and Soda Affect Cancer Patients
Navigating the Sweet and Bubbly as a Cancer Patient Dr. Urvi Shah Shares the Impact of Sugar and Soda on Cancer Patients As you navigate your cancer journey, questions about diet often bubble to the surface. Dr. Urvi Shah, is a board-certified hematologist-oncologist at Memorial Sloan Kettering and a Hodkin lymphoma patient. Dr. Shah led…
-

·
How Long Can You Live with Chronic Lymphocytic Leukemia
How Long Can You Live with Chronic Lymphocytic Leukemia? Dr. Adam Kittai and Dr. Joanna Rhodes share their thoughts on CLL life expectancy Receiving a chronic lymphocytic leukemia (CLL) diagnosis is likely to lead to a very human question: How long can I or my loved one live with CLL? To answer that question, we…
-
·
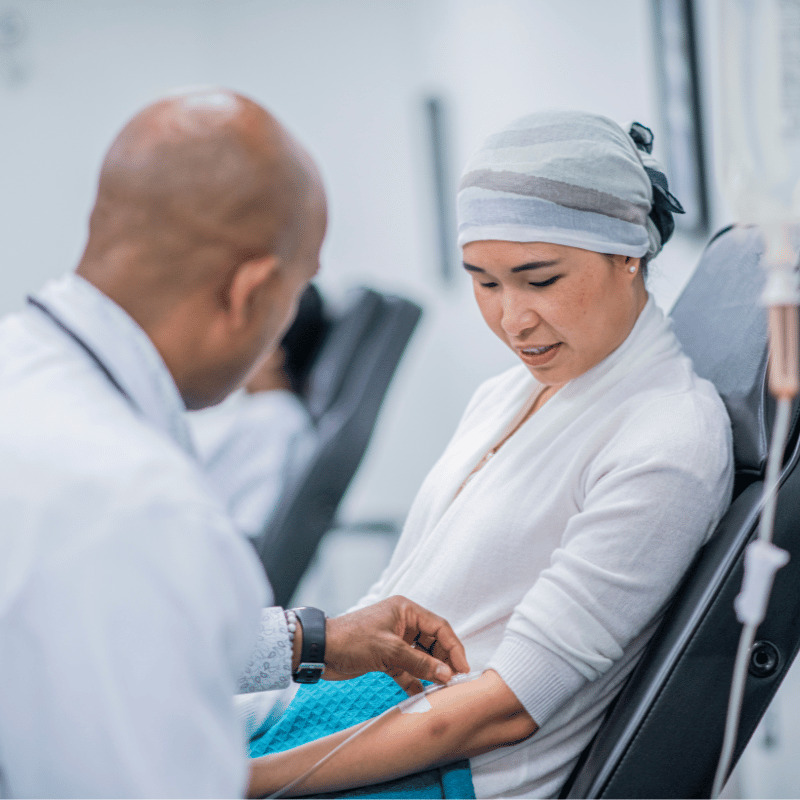
Significant Disparity: Myeloma Real-World Results Show Striking Contrast with Clinical Trials
Worse Outcomes for Myeloma Patients in Real-World Results vs. Clinical Trials Multiple myeloma patient outcomes are strikingly different in the real-world versus controlled environments according to data presented at the ASH Conference 2023 (Abstract 541). In the real world, multiple myeloma patients demonstrated a 44% reduction in progression-free survival (PFS) and a 75% decrease in…
-

·
Should Cancer Patients Reconsider CAR T-cell Therapy?
Should Cancer Patients Reconsider CAR T-cell Therapy? Explore CAR T-cell therapy insights with Dr. Joshua Brody amid FDA investigation. The headlines about the recent FDA investigation into CAR T-cell therapy have raised questions about the cancer treatment for many patients. To provide some answers, we sat down with Dr. Joshua Brody, Director of the Lymphoma…