The Latest in Multiple Myeloma Treatments
What Clinical Trials are Available to Me?
Edited by:
Katrina Villareal
Led by patient advocate Cindy Chmielewski, this enlightening session features experts Dr. Carl Ola Landgren (Sylvester Comprehensive Cancer Center, University of Miami) and Dr. Caitlin Costello (University of California San Diego). Together with Clinical Trial Nurse Christen Hawthorne (The Leukemia & Lymphoma Society), they will uncover groundbreaking insights
Learn about new multiple myeloma treatment classes and combinations, including advances in BCMA-targeted therapies, like CAR T-cell therapy and bispecific antibodies. Find out the future of belantamab mafodotin combinations in relapsed/refractory multiple myeloma. Acquire clinical trial knowledge and bust common misconceptions about clinical trials.

Brought to you in partnership with The Leukemia & Lymphoma Society and its Clinical Trial Support Center.

Thank you to GSK for its support of our patient education program! The Patient Story retains full editorial control over all content.
This interview has been edited for clarity and length. This is not medical advice. Please consult with your healthcare provider for treatment decisions.
- Introduction
- What is Multiple Myeloma?
- Common Symptoms of Myeloma
- Navigating Myeloma Treatment Options
- Working Toward Cure
- Trial Knowledge #1: Phases
- Trial Knowledge #2: Placebo
- Trial Knowledge #3: Finding Clinical Trials
- Clinical Trials for Newly Diagnosed Patients
- Triple-Drug Combination Clinical Trials
- Four-Drug Combinations for Multiple Myeloma
- CAR T-cell Therapy
- CARTITUDE trials
- Bispecific antibodies
- Choosing Between CAR T-cell Therapy & Bispecific Antibodies
- The Role of Stem Cell Transplant
- Final Takeaways
Introduction
Stephanie Chuang, The Patient Story: My name is Stephanie Chuang, founder of The Patient Story. A few years ago, I had a diagnosis of non-Hodgkin lymphoma, but we have so many shared experiences in what we’re looking for.
The Patient Story tries to help both patients and care partners navigate life after a cancer diagnosis and we do this primarily through in-depth conversations with patients, care partners, caregivers, and top specialists across cancers.
We believe it’s especially important to have these educational programs specifically for patients, family members, and supporters when it comes to topics like clinical trials, which can sound daunting and have lots of jargon so our goal is to humanize your options.
We’re so proud to be co-hosting this with The Leukemia & Lymphoma Society, a great partner of ours. It’s the world’s largest nonprofit health organization dedicated to funding blood cancer research that is so critical as well as offers patient services and education.
They have great resources, including information specialists who are a phone call away to help answer your cancer questions. We’ll be highlighting the LLS’ Clinical Trial Support Center or CTSC, which is a huge free resource that offers one-on-one support to enroll and stay in clinical trials.

We also want to say a special thanks to GSK for supporting our educational program. Their support helps us make these programs more available and free for our audience. We want to note that The Leukemia & Lymphoma Society and The Patient Story retain full editorial control of this entire program.
This is not meant to be a substitute for medical advice. We want you to walk away and be able to go to your own medical team with questions and more knowledge to make more empowered decisions.

Cindy Chmielewski, Myeloma Patient Advocate
Stephanie: Our first incredible panelist, a myeloma patient voice, and leader in the community, Cindy Chmielewski, will be leading this conversation from this point forward.
Cindy, I know you’re going to share more about your own story and how you became such a passionate advocate, but we would love to learn more about you because as we know, we are so much more than a diagnosis.
Cindy Chmielewski: Hi, everyone! My name is Cindy Chmielewski and I’ve been living with multiple myeloma since 2008. Prior to my myeloma diagnosis, I was a fifth-grade teacher.
Since being diagnosed with myeloma, I realized how important it is for patients to be part of their care and educated so they can be part of the decision-making process. Now I’m using my teaching skills to help myeloma patients learn more about myeloma so they can engage in discussions with their doctors.
Dr. Caitlin Costello, Hematologist-Oncologist
Cindy: We have Dr. Caitlin Costello, a hematologist-oncologist and associate professor of medicine at the University of California San Diego. She specializes in treating blood cancers with a focus on multiple myeloma. She also participates in a number of clinical trials that offer cutting-edge treatment strategies and therapies for myeloma.
Dr. Costello, what drew you to myeloma and what’s your passion for helping patients?

Dr. Caitlin Costello: It’s people like you who draw us into the things we love. Multiple myeloma satisfied a lot of my interests. The science of myeloma exploded during the course of my career, which really satisfies the academic side of my brain.
I didn’t want to go into oncology when I was doing my training. I find such passion and reward in connecting with my patients. At the very beginning of my training, I felt like cancer oncology was not going to allow me to have long-standing relationships with patients because of the biology of the disease and the outcomes associated with it.
I found myeloma and it satisfied both. It let me find that scientific side and let me have enduring relationships with patients as myeloma patients live long with this cancer. Sometimes things fall into place and I’m so grateful for that.
Cindy: I’m so glad that we are living much longer. We do develop relationships with our doctors because we’re in ongoing treatment so we’re seen for a very, very long period of time.
Dr. Carl Ola Landgren, Hematologist-Oncologist
Cindy: Dr. Carl Ola Landgren is a hematologist-oncologist at the University of Miami Sylvester Comprehensive Care Center. He is a professor of medicine, chief of the myeloma division, and co-leader of the Translational and Clinical Oncology Program. He also has a strong interest in the development of early treatment clinical trials.
Dr. Landgren, what drew you to myeloma, and what drives you most to continue your work with helping patients?
Dr. Carl Ola Landgren: I’ve been in the field for quite a few years; almost 30 years since I started my career.
When I was in fellowship, the senior doctors were saying there are two diseases you should stay away from, chronic lymphocytic leukemia and multiple myeloma, because there had been so little development in those disease areas.
I thought, That sounds like a really great opportunity. We need to go there. We need to do more investigation and see if we can change that. I was drawn to that and initially started working on both. Over time, I picked multiple myeloma as the disease that I was most interested in.
It’s been spectacular to be part of this journey together with so many other colleagues and friends around the world, with all the development of new drugs, and all the new technologies.
Over three decades, we are at a very different place. I’m very, very happy about it. All my research is centered on patient needs. I’m dedicated to driving the research forward.
Cindy: Thank you for choosing myeloma. We’re very lucky to have you because I’ve seen all the work that you do.

Christen Hawthorne, Clinical Trial Nurse Navigator
Cindy: I would like to introduce Christen Hawthorne from The Leukemia & Lymphoma Society, a nurse navigator with the Clinical Trial Support Center. Christen, share a little bit about yourself and what made you become a clinical trial nurse navigator.

Christen Hawthorne: I’m originally from northern New York, but moved to Denver, Colorado, in 2015 to pursue a career in cancer care. Since then, I’ve specialized in the area of bone marrow transplants and worked with patients who have a blood cancer diagnosis at different stages in their treatment plan.
My grandfather, unfortunately, passed away from cancer. I was so inspired by the holistic care that he received not only when he was in the hospital but also from my grandmother, who’s a retired registered nurse. Seeing that care and devotion, both professionally and personally, inspired me to pursue a specialty that allowed me to care for people beyond the clinical landscape and really focus on them as a person.
I’ve volunteered for The Leukemia & Lymphoma Society since 2015, fundraising for Light The Night and as an advocate with their Office of Public Policy. Seeing the many different pillars at LLS and how they support patients and families inspired me to pursue a position within the organization, joining the CTSC team in 2022.
Clinical trials and research are critical pillars of cancer care. They are how efficacious treatments are developed with the hope of fewer side effects and better outcomes. I hope I’m able to shed some light on their importance, dispel myths, and share additional resources.
Cindy: The myeloma landscape has changed over the last decade or two for clinicians, researchers, and patients.
When I was diagnosed in 2008, there were very, very few treatment options. Most of what’s available today were not available. Induction therapy was normally a doublet. Eventually, it became a triplet. Now we have quadruplets.
Back in 2015, there was something called the November to Remember and it was in that November when three myeloma drugs got approved within weeks of each other. We had our first two monoclonal antibodies and an oral proteasome inhibitor. It seemed like a great time.
But since then, we’ve had so many more drug approvals that I can’t even keep track of them — CAR T cells, bispecific antibodies, and the next generations of some of our other drugs. It really is an exciting time.
With all these drugs coming into the market, it’s so important that myeloma patients work with the myeloma specialists because there are so many combinations. How do you sequence them? What’s best for you? Are there any biomarkers that will help predict what would be a better outcome for you?
The landscape is changing, which is exciting, but we have a lot of work to do to figure out the best combination for each patient.

Multiple myeloma is technically a cancer of a cell called a plasma cell, which is an important part of your immune system.
Dr. Caitlin Costello
What is Multiple Myeloma?
Cindy: Dr. Costello, can you briefly describe what multiple myeloma is and some of the signs and symptoms of multiple myeloma?
Dr. Costello: It’s so important to do myeloma 101. Patients can advocate for themselves if they understand the basics of the disease.
Multiple myeloma is technically a cancer of a cell called a plasma cell, which is an important part of your immune system.
When they do that, they don’t work like they’re supposed to either. Now you have cancerous plasma cells that are not working the way they’re supposed to and they start producing abnormal proteins.
Your body, your immune system, and your plasma cells are supposed to produce a variety of different proteins or antibodies to fight the bad guys. But when one plasma cell goes rogue and starts copying itself, it starts prioritizing to make one form of those antibodies relative to the rest of them. Those bad antibodies get produced and find a way to run around the body and wreak havoc a bit.
Significant fatigue, shortness of breath, and lightheadedness are some of the most common symptoms that patients come to get medical help for when they’re first diagnosed with myeloma.
Dr. Caitlin Costello
Common Symptoms of Myeloma
Dr. Costello: Myeloma can affect your body in a variety of ways, one of which is having an interest in the bones. There are a couple of different ways it affects the bones. I like to think of it as a little Pac-Man. It takes little bites out of the bones or can almost form little tumors within the bones and that can hurt.
A lot of patients who are first diagnosed with myeloma say, “I have this pain. I don’t know why it’s there and it’s not going away,” and they seek care for that. Sometimes it can take a while to get to a diagnosis of multiple myeloma, but bone pain is a common presenting symptom.
Another common presentation is fatigue and this is hard because we’re all tired; life is hard sometimes. Fatigue can happen because of anemia.
Multiple myeloma is a problem in your bone marrow, which is the factory not only of your immune system but also of your blood. If your bone marrow is busy making cancerous cells, there’s not enough room to make the normal blood you need. That decreased production known as anemia can really produce symptoms of significant fatigue, shortness of breath, and lightheadedness.

Those are some of the most common symptoms that patients come to get medical help for when they’re first diagnosed with myeloma.
Cindy: They were the two symptoms that brought me to the doctor. I had terrible back pain and I was so tired and out of breath.
A big step forward in the field has been the evolution of all the different options. It’s no longer a matter of picking A or B. We pretty much have the whole alphabet to present.
Dr. Carl Ola Landgren
Navigating Myeloma Treatment Options
Cindy: We’ve had a slew of new treatments. When you think about what treatments a person should be on, what are some considerations that you weigh? What do you think about when they’re transplant eligible or ineligible, newly diagnosed versus relapsed/refractory?

Dr. Landgren: A big step forward in the field has been the evolution of all the different options. It’s no longer a matter of picking A or B. We pretty much have the whole alphabet to present.
There are very many options and it starts in the newly diagnosed setting. If someone has a recurrence, there are many options looking at the guidelines that dictate what we’re allowed to prescribe. Based on the NCCN Guidelines, there are over 40 different combinations in the setting of relapsed/refractory disease.
When I have a newly diagnosed patient, I have to think if this person is a potential transplant candidate or not. That has also shifted over the years because we can technically transplant many more patients than we could before. We can transplant people who are older and even those with comorbidities.
On the flip side, I personally think that the need for transplants has probably gone down. There are so many new treatments so we could do more. But in reality, I think we do less and I think the numbers point in that direction as well.
Cindy: I’m glad you include the patient. Shared decision-making is always the best.
We have to understand the difference between a long, durable remission, which means it goes away and stays away for some great period of time, versus a cure, which means it ain’t ever coming back, which is what we’re all aspiring to.
Dr. Caitlin Costello
Working Toward Cure
Cindy: Is there a treatment that can cure myeloma or put myeloma in a really deep remission? A few years ago, doctors were very hesitant to use the word cure. They were talking about controlling myeloma and keeping it at bay for several years, but I never heard the word cure. But more recently, I’ve heard the word cure and doctors are thinking that maybe we are curing some patients.
I talked to lots of patients and one of the most common questions I hear is what treatment works the best. Should I be on three drugs? Should I be on four drugs? What should I have? They also want to know: is there a cure for myeloma?
The words myeloma and cure were never used in the same sentence by myeloma specialists, but things began to change. I started hearing myeloma doctors say, “Yes, I think we may be curing some patients and maybe in 10 years, we’ll be curing more patients.”
Dr. Costello, can you talk about your perspective on a cure for myeloma?
Dr. Costello: This is the other C word. The big C we think about is cancer, but the cure word is becoming something we’re really starting to flirt with. Over the last couple of years, we’ve seen this explosion of new medications.
In the last two decades or so, we have seen the number of options for treatment go from a few to a wide variety of options that let people get into remission and stay in remission for very durable long periods of time. We’re starting to see that happening for such long periods of time that we’re saying, “Is it ever coming back?”
We have to understand the difference between a long, durable remission, which means it goes away and stays away for some great period of time, versus a cure, which means it ain’t ever coming back, which is what we’re all aspiring to.

I think we’re getting there. I always tell my patients, “In my lifetime, we’re going to do this. We absolutely are going to do this.” We’ve seen too much change in the last decade or two decades to not reach that point.
This is not a one-size-fits-all type of cancer. Although multiple myeloma is grouped together as one form of blood cancer, there are very different subtypes and very different forms of myeloma that act differently. Some are easier to treat and may go away, others can be a little bit more stubborn.
When we’re thinking about ideal treatments for any individual person, we have to look at you as a person, your health, your social situation, and your support. Then we have to look at the biology of the disease and put all of that together to figure out the best means of achieving a cure.
With more and more of these drugs delivering great, deep, durable responses, it’s natural for the field to talk about it.
Dr. Carl Ola Landgren
Dr. Landgren: I do think we are heading towards a cure. There are multiple reasons that the field has not talked about that cure for myeloma in the past. Quite frankly, the drugs were not good enough. We used to have 1 or 2 drugs for many years, way before I started practicing.
Then there was the emergence of high-dose chemotherapy with melphalan, the so-called transplant. Then there was the rediscovery of IMiDs with Dr. Bart Barlogie’s discovery of thalidomide that led to lenalidomide and pomalidomide. We had proteasome inhibitors coming around. Then we had all the immunotherapy.
The field has evolved. We see many patients achieving remissions and minimal residual disease (MRD) negativity so there is no detectable disease. When you don’t have those perspectives, it’s not really something brought up frequently with cure. With more and more of these drugs delivering great, deep, durable responses, it’s natural for the field to talk about it.

There is really no definition in the literature. What is the definition of cure? It’s true for any disease. If you were to survey myeloma experts and ask how they define cure, you would probably get very different answers.
If you ask me what I think, someone who has been treated with effective therapy and repeatedly shows no detectable disease is, at some point, probably cured.
If you put me on the spot and say how long that window is, then I will start going back and forth but maybe five years or so is a reasonable endpoint. Can you be 100% sure that the disease will never come back? Probably not, but life is filled with a lot of unknowns.
Many patients in my clinic are cured. Many of those patients come from Dr. Barlogie, who used to treat a lot of patients in Arkansas. He gave every drug under the sun. When he moved to New York, I was the chief of the Sloan Kettering myeloma program then he retired. When he retired, many of those patients were looking for another doctor and came to me. Then I moved to Florida. Many of those patients followed me to Miami.
I had many of his former patients who were treated for five years and then stopped treatment. I have patients who have been off treatment for 20 years. They remain off treatment and are doing very, very well.
Cindy: Thanks for giving me hope that you think there’ll be a cure in my lifetime so that makes me smile.
All drugs have to move through steps or phases to ultimately be approved by the FDA.
Christen Hawthorne
Trial Knowledge #1: Phases
Cindy: We have a new section called Trial Knowledge where we’re going to give you some knowledge about clinical trials and maybe do some myth-busting of things we’ve heard.
Christen, there are different phases of clinical trials. Can you talk about what happens in each of those phases?
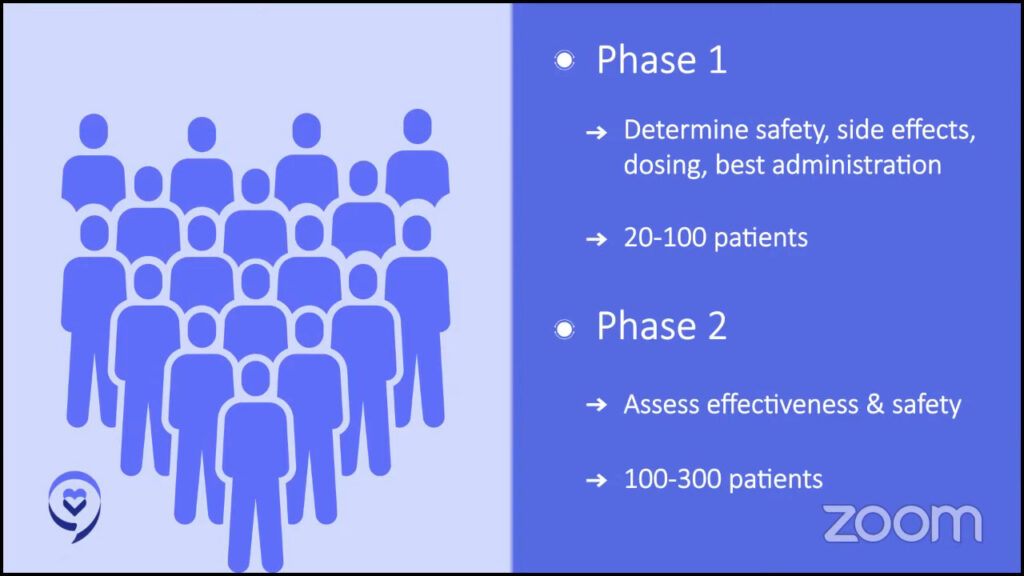
Christen: I like to think of clinical trials as steps to drug approval. All drugs have to move through steps or phases to ultimately be approved by the FDA. If drugs are studied in new combinations or in diseases outside of the approved indication of the drug, they still have to go through these phases.
In phase 1, we’re investigating the safety, side effects, dosing, and best way to administer treatment. This is usually in 20 to 100 patients.
In phase 2, we’re determining the effectiveness and safety of the study drug in about 100 to 300 patients.
In phase 3, we’re looking at the effectiveness, side effects, and safety of the study drug in comparison with other treatments. This includes anywhere from a few hundred to a couple thousand patients.


No one with active cancer would be treated with only a placebo as that is unethical.
Christen Hawthorne
Trial Knowledge #2: Placebo
Cindy: One thing I hear from lots of patients and care partners is that they really don’t want to consider clinical trials because they’re afraid that they’re going to be given a placebo. With cancer, you don’t want to be given a placebo. Can you talk about placebos and how they are used in cancer trials?
Christen: It’s definitely a common one that we get from our patients.
Federal regulations require patients to know if a placebo or a substance that looks the same as the one used in the treatment but is inactive will be used in a trial. Placebos are not typically used with patients in cancer care trials. The decision of whether to use a placebo in a clinical trial is based on how serious the illness is or if the disease is life-threatening.
Patients with these types of diseases are given the best available treatment or standard of care instead of a placebo. If used in cancer clinical trials, the placebo will be in combination with active drugs so you’re at least receiving the standard of care. No one with active cancer would be treated with only a placebo as that is unethical.

Cindy: It’s good to know that if there is a placebo, it’s in addition to the standard of care that you normally would be receiving. You will never be given just a placebo alone.
Clinical trials are a very complicated care landscape to navigate. The mission of the Clinical Trial Support Center is to help patients and caregivers identify potential clinical trials and overcome the barriers to enrollment.
Christen Hawthorne
Trial Knowledge #3: Finding Clinical Trials
Cindy: Christen, how do patients find clinical trials? I know there’s ClinicalTrials.gov, but it’s not the most user-friendly. You might end up with hundreds of trials when you search for them and you won’t know what’s right for you. Can you talk about how The Leukemia & Lymphoma Society can help?
Christen: Clinical trials are a very complicated care landscape to navigate between understanding what institutions are participating in certain trials, their inclusion and exclusion criteria, and what might be the best match for your unique profile. It can be really overwhelming.
The national average of cancer clinical trial enrollment is only between 5 and 10%. We found that, on average, our team has 22 interactions on behalf of a patient from the initial assessment to the time of enrollment — that’s 22 potential holes in the road where patients or caregivers could fall through. We’re able to fill those holes in 23% of eligible patients that we work with enroll in a clinical trial.

Resources within the LLS, including the Clinical Trial Support Center, collaborate with your care team, helping equip you with the knowledge to support you and to help you feel confident in making an informed decision about your care.
Our support center is a team of nurses and nurse practitioners who work one-on-one with you and your family to understand your unique clinical situation while getting to know you as a human, providing support that aligns with your goals of care.
Our mission is to help patients and caregivers identify potential clinical trials and overcome the barriers to enrollment.
We start with an initial conversation to learn more about you, your goals of care, your treatment plan, and your past medical history. From there, we begin a search based on the information provided, including ability and willingness to travel, insurance concerns, and any other factors specific to you.
Once our search is complete, we provide a comprehensive but easy-to-digest list of potential trials and resources to review with your care team. If there is an appropriate trial to pursue, we can reach out to the clinical trial sponsor or specific site with any questions or find out the next steps to enroll on your behalf. Throughout this process, we remain available to support you and answer questions along the way.
You can connect with one of our information resource specialists. They are happy to assist you in filling out the form and we can connect to get to know you and do a thorough assessment.
Cindy: Sometimes staying on a clinical trial may be difficult. Can you explain some of the things that the LLS can do to help patients stay on a trial?
Christen: In the Clinical Trial Support Center, we’re able to understand your unique clinical situation before providing a comprehensive list of trials to review with your care team. We also provide continuity of care by working with you and your family throughout your diagnosis and this could be months to years long. We continue to understand your evolving goals and tailor our support to meet them.
We do have regular meetings with sponsors of clinical trials to understand patient requirements of the trial and if there are additional support opportunities for patients or families to cover travel and lodging expenses associated with trial participation.
While we provide education and resources to our patients within the CTSC, we also have an incredible network of information resource specialists, the financial assistance department, and patient and community outreach managers who can help support patients throughout their care plans in unique ways.

Clinical trials exist at every part of the journey with myeloma and they really offer opportunities for patients.
Dr. Caitlin Costello
Clinical Trials for Newly Diagnosed Patients
Cindy: A newly diagnosed patient is someone who’s just starting treatment. In my mind, I always thought that clinical trials would be for later down the line when you’re running out of treatment options.
Dr. Costello, can you talk about trials for newly diagnosed patients?
Dr. Costello: Clinical trials exist at every part of the journey with myeloma and they really offer opportunities for patients. Like Christen beautifully said, you’re either getting the standard of care or you’re getting something that might be better so it gives patients opportunities for the next new and cutting-edge treatments.
In the last few years, we’ve had the outcomes of some of these newly diagnosed trials, particularly the ones that finally are getting published. One was the GRIFFIN study. As Cindy mentioned previously, two drugs are good, three drugs look better so maybe four is the best.
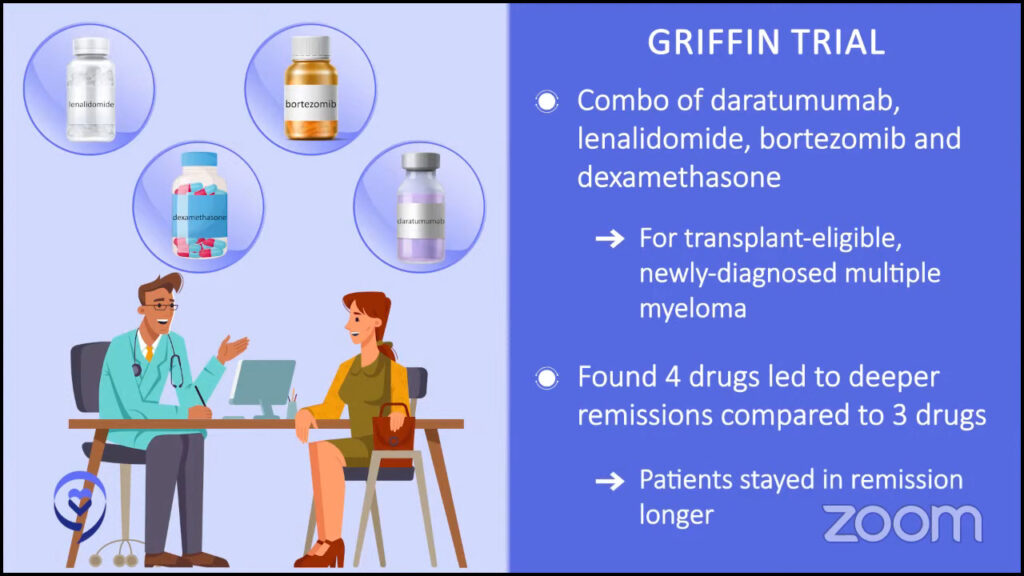
When someone’s newly diagnosed, our usual cocktail is three different drugs together. But what if we give them four? Does that have better outcomes and more likelihood of achieving remission? Does it get us more durable remissions where people are staying in remission longer?
We have the final analysis published that showed us that those patients who get four drugs when they’re first diagnosed have deeper and more durable remissions than those who get standard of care. Our standard of care of three drugs now seemed outdated.
It’s in recent years that we’ve said four drugs are the way to go. This is specific to younger and/or healthier patients. Age is just a number. The reality is some intensive therapies may be too much for a patient depending on their health or other medical issues that they may have.
If we find the next new and exciting thing for someone younger and/or healthier, the next question is: what about someone who may not be as young or as fit or possibly more frail?
The triplet combination that came out of the MAIA study combined three different drugs, which we proved was better than two different drugs. To say that, though, that cocktail truly has exceedingly, exceedingly effective outcomes for patients who may be older or more frail, for example.
There’s a lot of work that’s been done to get us to this point. We’re so glad that we finally have a new standard of care that we can have future trials use as a baseline to compare to for the next steps to get us to that cure.

Cindy: We’re in exciting times. If you ask different myeloma specialists, they would have different answers about what a cure is. The same holds true for patients. If you ask what they expect in a cure, you might come up with different answers. But to me, it sounds wonderful being treated for a set period of time and then being off drugs for an extended period of time; that would be my definition of a cure.
We need more classes of drugs because if you treat the patient and it works, that’s fantastic. But if the therapy stops working, we need to have other classes of drugs.
Dr. Carl Ola Landgren
Triple-Drug Combination Clinical Trials
Cindy: We’ve been talking about how things have changed. I know when I was diagnosed back in 2008, induction therapy was a doublet. Now, the standard of care is a triplet, but it seems to be moving towards quadruplets.
Before we start talking about quads and the benefits of some of these four-drug combinations, can we focus on some of the clinical trials that are triplet combinations and which ones have been gaining your attention and why?
Dr. Landgren: The bigger triplets that I pay attention to are the RVd or VRd regimen. This is bortezomib, lenalidomide, and dexamethasone. This was initially developed by Paul Richardson and the group at Dana-Farber. It was presented at ASH 2008 and it was published in a small study in 2010 that became the standard in the US, based on a little bit more than 50 patients treated.
Eventually, it was published as a randomized study compared to lenalidomide and dexamethasone. The randomized evidence came many, many years after this had already become a standard of care. That is one of the three-drug combinations that I think are important. They pushed the field forward.

I was involved in the development of the KRd regimen with carfilzomib, lenalidomide, and dexamethasone. The difference between these two regimens is that the proteasome inhibitors are different. You have no added neuropathy with carfilzomib, which you have with bortezomib.
But if you give carfilzomib to a patient with underlying cardiovascular disease, you could push that patient into congestive heart failure. What has been found also is that a lot of this is due to giving too much fluid. But that’s a great, great combination. For patients who are otherwise fit, that would be my go-to therapy.
Then you have the DRd, which is daratumumab or DARZALEX with lenalidomide and dexamethasone. That was approved in 2019. That has been a game-changer. It takes away all the problems with bortezomib’s neuropathy. You replace the proteasome inhibitor with an antibody. Giving a CD38-targeted antibody with lenalidomide and dexamethasone has really changed the field.
It has also erased some of the age discrimination that’s been in the field for a long time where younger patients could get more effective therapies while patients who were older and more frail have very few options. You had only two drugs.
With daratumumab coming, the difference between patients that are frailer, older, or have other issues and patients who are younger and more fit became smaller in terms of efficacious drugs. The addition of immunotherapy has really, really made a huge step forward.
Cindy: How about your thoughts on the trials that are doing selinexor, pomalidomide, and dexamethasone against elotuzumab, pomalidomide, and dexamethasone? Any thoughts on that trial?
Dr. Landgren: Those are combinations in trials for patients who have relapsed/refractory disease. Elotuzumab is a drug that binds to a protein called CS1 or SLAMF7, and it was discovered many years ago.
In the laboratory, this antibody bound to myeloma cells and they died. However, in patients, elotuzumab alone has not been found to have any clinical benefit.
Elotuzumab has been partnered either with lenalidomide or pomalidomide. Given in combination, it has been found to be more efficacious than lenalidomide with dexamethasone alone or pomalidomide with dexamethasone alone. Elotuzumab adds to these backbones.
Selinexor can be used in different combinations. It has been approved with bortezomib and for patients with a first, second, or third relapse. There are other combinations as well with pomalidomide and also with carfilzomib.
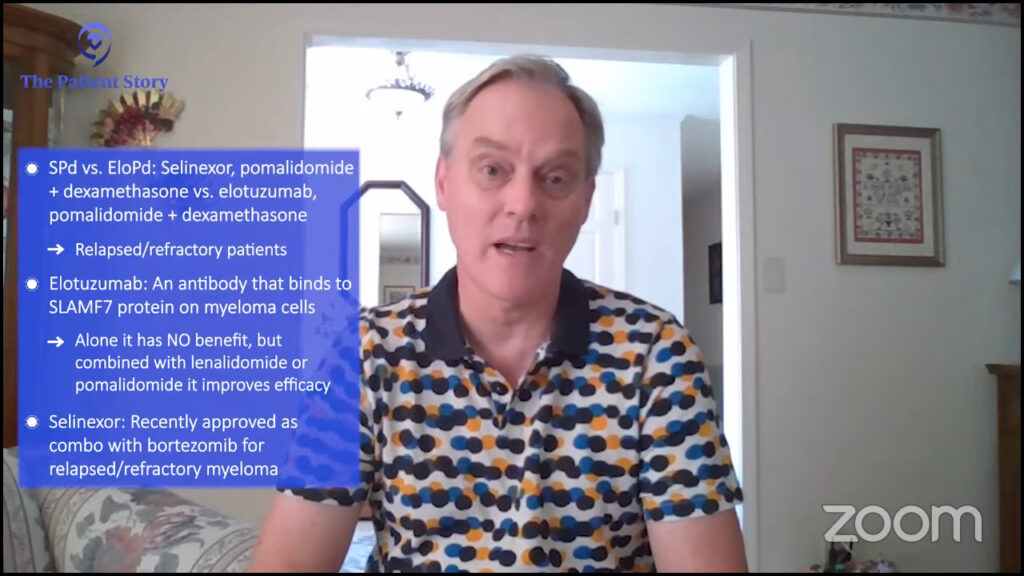
Selinexor is a new class of drugs. It binds to a protein that is sent into the center of the tumor cells, the nucleus, so it’s a blocker of certain proteins that go in and out of the nucleus and has been found to work very well in myeloma.
We need more classes of drugs because if you treat the patient and it works, that’s fantastic. But if the therapy stops working, we need to have other classes of drugs.
Cindy: Before we move on to the four-drug combinations, let’s talk about BLENREP or belantamab mafodotin. We had it on the market for a bit and then it was pulled because it didn’t meet one of its primary endpoints, but it’s still in trials, like the DREAMM 8 trial, using BLENREP in combination.

Dr. Landgren: Belantamab mafodotin is an antibody-drug conjugate. It’s a small molecule, a toxin similar to lenalidomide or carfilzomib that can have an anti-myeloma effect. The difference between belantamab mafodotin and these other drugs is the way it’s delivered.
The drug is not given as a tablet, an injection, or an infusion; it’s bound to an antibody and that antibody is infused into the vein. When the antibody comes into the human body, it looks for the corresponding protein that this antibody’s been designed to bind to and that is the BCMA protein that’s widely expressed on the surface of myeloma cells.
When these antibodies find the myeloma cells, they bind to the BCMA protein. Once they have, the myeloma cell lets this antibody in and once it’s in there, it releases this toxin that will kill the myeloma cells. It’s like a Trojan horse.
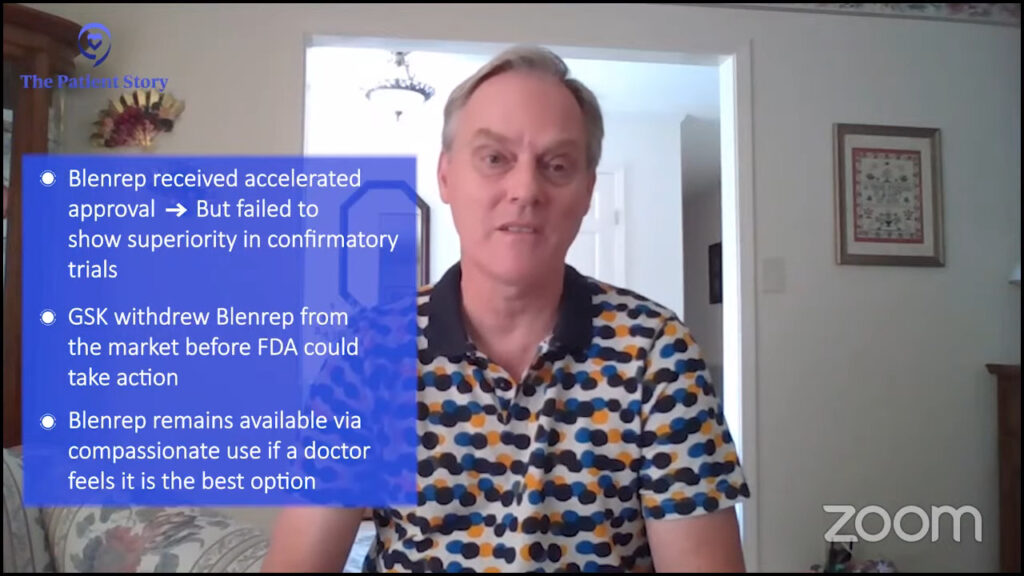
This is the first and so far only antibody-drug conjugate developed for myeloma. It was approved in 2020 and then pulled from the market in 2022. What happened?
The FDA has a fast track for drug development, which is great for patients. That will speed up the availability of new drugs. A drug company can do all the safety and toxicity data pre-clinically. They can open a phase 1 study and identify the right dose, then you can have an expansion cohort or a phase 2 study and treat a number of patients per agreement with the FDA.
If the right proportion of patients responds to the drug, if the FDA agrees, then an accelerated approval will happen. That’s what happened to most of the drugs. All the drugs we currently have in more recent times have been approved based on this mechanism; that happened to belantamab mafodotin.
When belantamab mafodotin was compared to its control arm, the drug belantamab mafodotin was not superior. When the FDA reviewed the data, they said this study did not live up to its endpoint.
FDA did not stop the drug. GSK, who owns the drug, took it off the market before the FDA made that decision. There was nothing for the FDA to stop because they already took it off the market.
However, in agreement with the FDA, the drug is available for compassionate use. The fact is that the drug works for many patients. If there is a doctor who thinks this is the best option for a given patient here and now, that doctor can reach out to the FDA and get permission to use it.
The FDA has the next rule in place. Once you’re given accelerated approval, you have to design a randomized study where you prove that this drug is superior to a control arm. A randomized study has to be written, launched, and concluded, and the data has to be handed to the FDA.
There are other trials that they had already launched in combination with belantamab mafodotin and there are two of them ongoing. There is a control arm and now it’s belantamab mafodotin in combination.
The study that failed was belantamab mafodotin as a single drug versus a control arm. There are two ongoing trials that use combination therapies versus control arms. When they are done, the data will be handed back to the FDA.
It’s very likely that those trials, one or both of them, may read out with the drug being superior and that could reintroduce the drug and they could get the full approval. We have to just wait and see.

They are part of the DREAMM program. Belantamab mafodotin was initially developed in DREAMM 1 and then there’s been 2, 3, and 4, and so forth. Now we’re up to DREAMM 7 and DREAMM 8.
The trials use belantamab mafodotin. One of them uses it with bortezomib and dexamethasone and the control arm is bortezomib and dexamethasone. The other one uses it with pomalidomide and dexamethasone and the control arm is pomalidomide and dexamethasone.
With all things being equal, I do think that four drugs are here to stay. The GRIFFIN study showed us that it is better than three.
Dr. Caitlin Costello
Four-Drug Combinations for Multiple Myeloma
Cindy: Do you think the four-drug combinations are going to be the new standard of care for newly diagnosed myeloma? If so, which four drugs do you envision to be used as part of induction therapy? How do you weigh the risks and benefits of adding that other drug with the side effects it may cause? Would there be some patients that maybe could still use three?
Dr. Costello: Four drugs are definitely here to stay but not for everyone. We talked about the importance of personalizing this approach and how we take into consideration so many different aspects of you, your health, and your myeloma when we make these decisions.
With all things being equal, I do think that four drugs are here to stay. The GRIFFIN study showed us that it is better than three.
There are more trials coming out. GRIFFIN was a phase 2 trial, which showed us important things, but we want to extrapolate that data to use it in several hundred different patients to prove what was shown in the GRIFFIN study.
The next studies that are coming along, like the PERSEUS phase 3 trial, are going to show us how we can get the durability of response and prove that the initial study, GRIFFIN, was right and is proof that quadruplet or four drugs is here to stay.

Dr. Landgren: Similar to most things in life, there will be early adopters and people who will be the very last people to change their minds. Some people build their career on saying no, no, no all the time and that’s how the field goes.
The three-drug combinations I mentioned before are proteasome inhibitors with IMiDs and steroids, and the evolution has also been CD38-targeted antibodies with IMiDs and steroids.
When we talk about four-drug combinations, it’s basically a merge of those so that means that you add a CD38-targeted monoclonal antibody. For the most part, the studies out there are using daratumumab added to a proteasome inhibitor, IMiD, and steroid. In plain language, you’re talking about Dara-RVd or Dara-KRd; those are the two that have been presented.
There are three trials that have been published so far using those combinations. The first was the Dara-RVd randomized phase 2 study that was published about three years ago in the Blood journal. They wrote the protocol and the lead author is Peter Voorhees.
That had about 100 patients with daratumumab with RVd and about 100 patients just with RVd. Patients receive four cycles of either of the regimens. They were all transplanted then they got two more cycles with the same regimen that they started off with.
After completing the sixth cycle, the study looked to see if there was an improvement in the stringent complete response rate. There was a slightly higher stringent complete response rate if you add the daratumumab therefore the study was deemed successful. Clinically, you could argue: is that really meaningful? But that’s how the study was designed.
Patients who got dara-RVd continued on daratumumab with lenalidomide maintenance for two years. The patients who got RVd got lenalidomide maintenance.
What the study continued to show is that the rate of MRD negativity is much, much higher in the group that started and continued on daratumumab. They have plotted out the progression-free survival curves. The progression-free survival seems to be way superior in the four-drug combination.
But it’s not only the four-drug combination upfront that’s different between the two arms. It’s not four versus three. It’s also the maintenance being very, very different. You could ask the question: what would happen if you gave everybody three drugs to begin with and had a two-drug or one-drug maintenance, would that make a difference? That study has not been done so we don’t know the answer to that.

The other combination is Dara-KRd and I was the lead investigator in the first trial to be published called the MANHATTAN study. I did that when I was at Sloan Kettering. We gave Dara-KRd for eight cycles and patients were not transplanted in that study.
After four cycles of treatment, a patient had the option of collecting their stem cells or not, and after eight cycles, they had the option of being transplanted or not. Some patients were MRD negative and they chose to go for transplant.
The majority of patients who were MRD negative chose not to be transplanted so they kept their stem cells in the freezer. We have kept on following these patients. So far, we have not really seen any significant difference in whether you were transplanted or not, but the study was not designed to fully answer this question.
The third study that used Dara-KRd is the MASTER trial, which was developed by Luciano Costa. He treated a smaller number of patients. Patients got four cycles of Dara-KRd, were transplanted, and if they were MRD negative after each of these two steps, were taken off therapy
If they turned MRD negative, they got another four cycles of Dara-KRd and, again, if they were negative two times in a row, they were taken off therapy. They could get an additional four cycles so they could end up with a total of 12 cycles. Every patient was transplanted in this study.
He has shown that with a relatively short follow-up, patients who didn’t have adverse cytogenetics continued to be free from disease after 1 or 2 or so years, while patients who had more aggressive disease would have their disease flare back again if you take them off therapy. That’s where the field is right now.
From what we’ve seen so far, it’s been a new, wonderful treatment option for myeloma patients.
Dr. Caitlin Costello
CAR T-cell Therapy
Cindy: When I first heard about CAR T-cell therapy, it was when the University of Pennsylvania and CHOP had their first CAR T-cell patient and how well that response was. Can you talk about what CAR T-cell therapy is and some of the benefits of using this type of immunotherapy in treating myeloma? What could some of the challenges be?
Dr. Costello: This has been one of the single most exciting treatments I have seen in my career. They were developing CAR Ts for other kinds of cancer patients and none of us really knew what to expect. But over the course of the last decade or so, we’ve learned so much.
CAR T stands for chimeric antigen receptor T-cell therapy. Your T cells are part of your immune system. They are these fighter cells that are designed to look for bad guys that come into your presence, whatever that may be.
What if there was an opportunity to train your T cells to specifically look for your myeloma? Speaking of personalized medicine, it doesn’t get much better than this. This is your own immune system fighting your own cancer.
Every myeloma cell has little tags on it that identify it as a myeloma cell. There’s a tag called BCMA, B-cell maturation antigen, on the myeloma cell. CAR T cell is a way that we can train your T cells to look for that tag.
The logistics of it can be a little overwhelming and it’s one of the challenges of CAR T. If you’ve ever undergone a stem cell transplant or have heard of it, it feels very similar.
Your T cells are removed from your body, similar to a blood donation. They’re sent off to a specific company that re-engineers them in order to create and train the T cell to look for that tag we talked about.
The CAR T cells are mailed back and, with some additional logistics, your doctor will put them right back into your body. Your immune system is now trained to look for your myeloma cells.
There are two different CAR Ts that were approved by the FDA in 2023 for treating relapsed or relapsed/refractory multiple myeloma. It’s approved for patients who have had four or more different treatments.
The benefit of it is that it is a one-and-done treatment. Myeloma patients have probably been on myeloma treatments for a very long time. They keep a lid on everything. Maybe it can delay it from coming back. But with that comes cost, time, side effects, and a lot of additional challenges.

When you receive CAR T-cell therapy, that is the treatment, and your immune system is now trained to go to war against your myeloma and knock everything down. It’s a very exciting opportunity to allow patients to have a treatment-free interval where they’ve done their treatment and can enjoy years without any additional therapy.
Unfortunately, it’s not curing everybody yet, but it’s buying us time, which is what we really do with myeloma treatments. We kick the can down the road until the next treatment comes.
There are lots of exciting benefits. With new drugs come new side effects that your doctor and you need to learn about and figure out how to manage to keep you safe. With new exciting options, there’s a lot of popularity around them.
There are a lot of logistics involved in getting the T cells out of you, engineering them, sending them back to you, having the manufacturing capabilities to do that, and that is time, money, and accessibility.
There is still not enough access for everybody who needs it to have it. We’re going to continue to fine-tune and hone this. But from what we’ve seen so far, it’s been a new, wonderful treatment option for myeloma patients.
Cindy: Patients were so excited when we had our first approved CAR T-cell therapy. It prolonged life. People who were out of options were given an option. That’s what we do in myeloma. We just keep on treating ’til the next new thing.
These CARTITUDE studies are looking to see if CAR T-cell therapy is at least as good, if not better, than our standards of care.
Dr. Caitlin Costello
CARTITUDE trials
Cindy: The CARTITUDE trials talked about cilta-cel or CARVYKTI. Can you talk a little bit about what we found out from the CARTITUDE trials and maybe specifically CARTITUDE-4 and what’s happening in CARTITUDE-5 and 6?
Dr. Costello: The CARTITUDE trials are looking at how can we use cilta-cel, which is one of the two CAR T products targeting BCMA.
Drugs get approved on the basis of safety and effectiveness from the very get-go. Truly, and maybe unfortunately, this is oftentimes studied with patients who have no other options.
Once we prove it works and that it is safe, then we can cast a wider net. If it works at that point in the disease, could we potentially study it in patients who have been diagnosed with myeloma more recently or have had fewer than four treatments or maybe even when they were first diagnosed?

The CARTITUDE trials, and there are a lot of them, are looking at exactly this. It’s a randomized trial that gives one set of patients the standard of care, what I know works, and another set of patients CAR T cells. These CARTITUDE studies are looking to see if CAR T-cell therapy is at least as good, if not better, than our standards of care.
We’ve been doing bone marrow transplants for a very long time and it has been great in the sense that it works. It has its issues, it has its side effects, and there are parts of it that are no fun. But everyone’s starting to say: can we compare that to CAR T instead?

We’re going to see more and more data coming out about some of these CARTITUDE studies. There’s the KarMMa study, which is evaluating the other BCMA CAR T cell with ide-cel, asking the age-old question: do we still need a transplant?
As a disclaimer, I’m a transplanter. I believe in transplants. Until someone shows me it’s better, there’s still a great role for transplant, but I can’t wait to see how those results turn out.
Cindy: I’m excited about that, too, because I haven’t had CAR T cell. I’ve had a transplant and from everyone I’ve talked to who’s had both, they had an easier time going through the CAR T-cell therapy.
The other more exciting thing is trying to put CAR T cells closer to the front line. As patients, we hear that sometimes CAR T cells aren’t working as great as they can because of exhausted T cells so if we did them sooner, they might not be exhausted. I’m hoping to see if that might be true and if we use some of these CAR T cell therapies upfront, maybe they’ll be closer to being curative than they are later on down the road.
The only way we’re going to find these answers is from clinical trials so we need them, we need patients to enroll in them, we need them to answer these questions, and we need those answers fast because patients are waiting.
We give an antibody that links the T cells to the myeloma cells. We are trying to engage the T cells so the T cells can act as the treatment for myeloma.
Dr. Carl Ola Landgren
Bispecific antibodies
Cindy: Bispecific antibodies are a new type of immunotherapy, too. Can you talk about what bispecific antibodies are, their mechanism of action, and some of the side effects that we’re seeing?
Dr. Landgren: From a development point of view, bispecific antibodies are very different from other antibodies. We talked about daratumumab and elotuzumab before they were developed back in 2015. The first bispecific antibody that came to myeloma was at the end of October 2022. We have additional two bispecifics in August 2023.
Bispecific antibodies bind to the surface of the myeloma cells and to the surface of T cells. They bring these two cells to sit next to each other. When a T cell sits next to the myeloma cell, it will kill the myeloma cell.
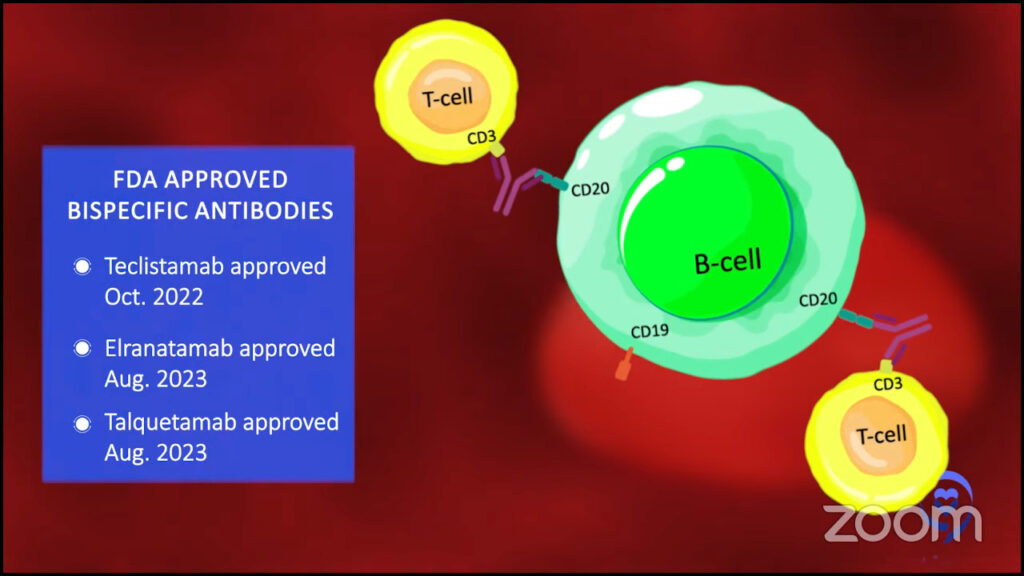
When we talk about CAR T-cell therapy, what does that mean? We take out the T cells, make them into CAR T cells, and those T cells bind to myeloma cells. You could say it’s very similar.
Instead of taking out the T cells, we keep the T cells in the body, but we give an antibody that links the T cells to the myeloma cells. We are trying to engage the T cells so the T cells can act as the treatment for myeloma.

The side effects of bispecific antibodies are quite similar to CAR T cells in that you could have cytokine release syndrome and these immune cell-mediated neurological syndromes with neurological side effects.
But usually with bispecific antibodies, you have less profound, less severe reactions. Most of the cytokine release syndrome we see are grade 1 or grade 2, but we very seldom or almost never see higher grades. But with CAR T cells, you could see grade 3 or grade 4.
There is some continued immunosuppression and patients seem to be more able to capture infections over time with bispecific antibodies.
Dr. Carl Ola Landgren
Cindy: Do bispecifics have higher rates of infection?
Dr. Landgren: Infections can happen both with CAR T cells and bispecific antibodies. I spent a lot of time going through the literature, trying to understand the dynamics of these types of immune cells and the side effects from both short- and long-term perspectives. I continuously go through the literature and talk to other investigators.
What I’ve seen is that the infections are more likely to happen earlier on with CAR T cells because you have more profound suppression. With the antibodies, the way they are developed is you keep on giving them. You not only hit the myeloma if it’s there or keep the myeloma away, but you also keep on hitting the immune system. You will see there is some continued immunosuppression and patients seem to be more able to capture infections over time with bispecific antibodies.

There are ways to prevent this and this all comes back to the physicians. Every time I talk about this, I always emphasize that doctors have to know exactly how to manage this.
These antibodies bind to markers, like BCMA and GPRC5D, which are widely expressed on myeloma cells. Unfortunately, these can be also expressed on normal plasma cells, which means that the patient would make fewer immunoglobulins. Low IgG in the body is called hypogammaglobulinemia and that could increase the risk for capturing infections. It could also be more severe infections.
A way to prevent these infections is to give intravenous immunoglobulin or IVIg. I always say to check your IgG levels. If they go under 400, you have to give IVIg. I would give it, check the next month, give it, and check the next month. If it goes into the normal range, then you can hold off, but you have to keep on checking it. If it starts drifting down again, you have to give it.
You can see with these combinations that you can bump up response rates from 60 to 70 to 80 to 90 to 95%.
Dr. Carl Ola Landgren
Cindy: The bispecifics that are currently approved are approved as monotherapy or just by themselves. There are some trials that are doing some combinations. Can you talk about those trials?
Dr. Landgren: The field is moving forward very fast and there is a whole range of combinations.
Damian Green of the Fred Hutch Cancer Center in Seattle found that myeloma cells can protect themselves by chopping off BCMA from their surface, the target for many of these bispecific antibodies and the currently available CAR T cells.
The drug binds to the target, but the target gets chopped off so it’s circulating free in the blood. The cancer cells are swimming by, waving to these receptors and the drug. That’s not a good scenario. How could you prevent that from happening?

Dr. Green and his colleagues found that this happens when the cells release γ (gamma) secretase. They used γ-secretase inhibition to stop this from happening. There are newer developments now and we have been part of this as well.
It’s probably overkill to give every patient this. For some patients, it may be the right thing to do, but in general, it’s probably not needed. That’s one line of investigation. We are not yet done, but that seems to be the way the field is going.
Then you could increase the efficacy of it. You could use other antibodies. You could use daratumumab in combination with bispecifics.
People have daratumumab and teclistamab. Let’s put them together and see what happens. People have used teclistamab with lenalidomide or elranatamab, which is also an approved BCMA bispecific antibody, together with lenalidomide and pomalidomide. You can start using it also with proteasome inhibitors, carfilzomib, or other drugs.
Why not take two of these bispecifics at the same time? You could take a BCMA and GPRC5D-targeted bispecific antibody and use that together. There is data coming out and ASH 2023 will present a lot of this data.
The bottom line is that these single drugs are amazing. About 60% of patients respond to bispecifics. To put it in context, when daratumumab was approved in 2015, it was 30%.
Patients today have been treated with many more drugs when they go on trials. Almost 10 years ago, there were virtually no drugs around. You could argue and say patients today are sicker than they were 10 years ago and it still delivers over 60% so that’s amazing.
You can see with these combinations that you can bump up response rates from 60 to 70 to 80 to 90 to 95%. There are some studies treating 28 patients and 27 of them respond. It makes me wonder how we can learn from that so we can take the last person over the goal line.
We will probably have, I would say, 95 to 100% response rates in later lines. These drugs will probably become part of front-line therapy. What we have known for a long time is that what works in later lines usually works better in earlier lines. If we have this discussion in five years, everything will have probably changed.

Cindy: Exciting times. I’m very, very hopeful. I hope to be here five years from now. When I was newly diagnosed, five years from now was really scary to me. Now with all these new drugs, I’m counting on being here and having a discussion with you in five years.
It scientifically makes sense for me to put CAR before BiTE, but I do think we have the reality of challenges with CAR T that don’t allow us to sequence it the way we would want to.
Dr. Caitlin Costello
Choosing Between CAR T-cell Therapy & Bispecific Antibodies
Cindy: How do I know whether or not I should be looking for a bispecific antibody or a CAR T-cell therapy? Dr. Costello, how do you have that discussion with your patients? I know it’s probably going to be very specific, but what are some of your guiding principles?
Dr. Costello: We have to break this down into the ideal world and the real world.
In an ideal world, everyone has access to everything. We are sitting in front of our patients, say A or B, and we can do A or B soon but that is not the real world.
The real world makes that difference for usually at least two reasons. People relapse differently. Some people have myeloma that grows back very slowly so we have time to organize the next treatment and think through the logistics. We can prepare ourselves or get on waitlists for CAR T because that’s the reality of CAR T.
However, other patients have myeloma that relapses very quickly. We may not have the benefit of time to think about our next steps, pursue treatments that may not be locally available, or be on a CAR T-cell therapy waitlist. We have to consider all these different scenarios to help make that decision.

In an ideal world, I like the idea of starting with CAR T. I would love for patients to have time to get off treatment and enjoy treatment-free intervals.
Bispecific therapies are a great way to keep using the immune system to keep pushing against the cancer cells, but they’re going to get tired, too.
If we continuously ask the immune system to fight, fight, fight, and then we try to take those immune system cells out and re-engineer them and ask them to go to a whole ’nother battle, they may not be as effective.
It scientifically makes sense for me to put CAR before BiTE, but I do think we have the reality of challenges with CAR T that don’t allow us to sequence it the way we would want to.
The other thing worth mentioning is that there’s this concept of a waiting period because we have different ways to attack the BCMA tag. As we attack it, we can see that the tag level in your bloodstream goes down as your myeloma gets killed.
At some point, it may regrow as your myeloma grows and we may need that BCMA to start growing back so we can attack it again. I don’t know if we know the answer yet about the ideal waiting period between doing sequential BCMA-targeted therapies, but it also does rationally make sense to me.
I think CAR T buys us more time to allow the T cells to do their thing then allow the BCMA to redevelop to allow us to target it again. It’s a little complicated because we don’t know.
No one really knows for sure. The jury is still out. There are so many questions that we don’t really have the answer to when it comes to this.
Dr. Carl Ola Landgren
Cindy: We could have a whole discussion on BCMA treatment, sequencing, and waiting periods. There’s so much going on.
Dr. Landgren: We talk about it at conferences. It always comes up and people talk about it internally. I get phone calls from private practice doctors asking for advice on how to think about it.
The short version is no one really knows for sure. The jury is still out. There are so many questions that we don’t really have the answer to when it comes to this.
If you look at the trials that use bispecific antibodies, some of those trials have allowed patients with prior exposure to BCMA-targeted therapies, like belantamab mafodotin or CAR T-cell therapy. If you look at the overall results, the numbers are not significantly different.

Similarly, there are CAR T cell studies that have been done that allowed people who have been exposed to prior BCMA-targeted therapy. Again, if you look at these response rates, they don’t look that different.
Based on the small numbers from available studies, if you do these so-called stratifications — you look at one study whether they have prior exposure or not, or do another study and flip it around from CAR T cell to bispecific or bispecific to CAR T cell — it doesn’t look that drastically different. There is no significant P value. There is no definitive answer. That’s why I started off by saying we really don’t know.
We have to be open-minded and we have to involve the patient. Every patient will have his or her own perspective.
Dr. Carl Ola Landgren
The Role of Stem Cell Transplant
Cindy: With all these newer treatment options, is a stem cell transplant still necessary?
Dr. Landgren: That is a very difficult question to answer because it is all about what you have as alternatives.
In a broader context, if you think about the whole world, transplant has been proven for a very long time. It was initially developed in 1983 for a very old treatment that is a spinoff of mustard gas, which is what melphalan is. It was mustard gas discovered taken in to become melphalan chemotherapy; that is the so-called transplant. It has been implemented around the world for all these years.

In many countries, there is limited access. There are very few treatment options for patients. Transplant remains a very, very good alternative. In many parts of the world, in 2023, this remains one of the most, if not the most, powerful therapies widely available so to take that away would be the wrong thing.
If you look at the United States, we are very lucky. We have so many treatment options. Is it the ultimate therapy for every patient? Do you have to have to do it? The answer is more complicated. It’s probably more towards maybe not.
If you ask someone who is a transplant specialist, that person will say, “It’s been around for a long time. I use it all the time. It’s a great therapy. It’s not a big deal. You should really do it.” I have respect for that and I see where they come from.
There are other people who will say, “There are so many new treatments. We have CAR T cells and bispecifics. There are so many alternatives. The patient is already MRD negative. Why don’t you keep the cells in the freezer and give the patient the option of doing a delayed transplant if needed?” I would agree with that, too. I have great respect for that opinion.
We have to be open-minded and we have to involve the patient. Every patient will have his or her own perspective.
Cindy: That’s why it’s so important to have educated patients who could be part of that conversation and make the decision about what they feel is best for them.
We don’t have a cure yet, but we can control the disease and provide a very good quality of life for many, many patients with these drugs.
Dr. Carl Ola Landgren
Final Takeaways
Cindy: Thank you all for such a wonderful and incredible discussion. If you had to leave our audience with one takeaway message, what would that be?
Dr. Costello: Advocate for yourself. There is so much that you need to understand about your cancer so that you can find your best, personalized pathway forward. With all these resources, you have no excuse to not do that.
Dr. Landgren: Myeloma is at a tipping point as a disease. If I put all my years of experience into perspective, it used to be a disease that was almost like a kiss of death. Now it’s a disease where the lifespan for many patients is getting very close to, if not identical, to the general population.
We don’t have a cure yet, but we can control the disease and provide a very good quality of life for many, many patients with these drugs.
Seek advice from experts. If you choose to be treated locally, the expert can always be looped in and give advice if there needs to be some changes and strategic considerations. Involve an expert when you’re newly diagnosed or if there are new decisions to be made. The future is very bright and we are always here to help you.
Cindy: I’m glad there’s a bright future. You just made this girl’s day because that’s not what I was told in 2008 so I’m glad times have changed.
Christen: I hope that you feel less intimidated by the clinical trial landscape and are encouraged to reach out to support teams like the Clinical Trial Support Center. We’re here to support you through your journey, whatever it may look like.
Cindy: It’s so important to be involved in your care. When I was newly diagnosed, I blindly followed my doctor’s orders. I grew up in that age of doctor knows best so whatever the doctor told me to do, that’s what I did without asking questions, without being part of that decision-making process. I’ve learned how it feels to be educated and empowered and part of that decision-making process.

Stephanie: Thank you so much, Cindy, for being our incredible patient advocate and moderator.
Thank you also to Drs. Landgren and Costello for the work that you do in the clinic and in research to help move the landscape of myeloma options and treatment forward, and for helping patients and families who are trying to navigate a multiple myeloma diagnosis and treatment.
Thank you to our LLS clinical trial nurse, Christen Hawthorne, for being there for people who are overwhelmed by approaching clinical trials.
We really hope that you walk away with a better understanding of clinical trials, specifically in multiple myeloma. Again, they may or may not be right for you, but our goal is to make sure you feel empowered to make a decision for yourself. We hope to see you at a future program.

Special thanks again to GSK for its support of our independent patient education content. The Patient Story retains full editorial control.





